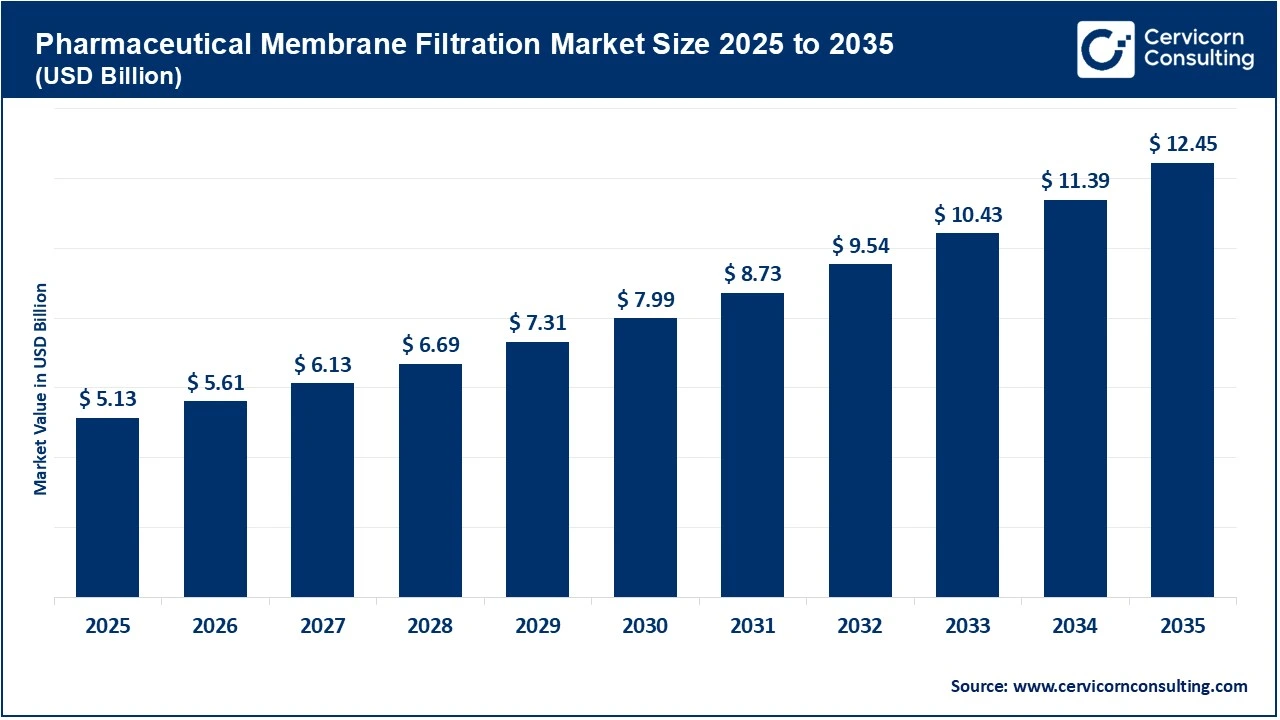
The global pharmaceutical membrane filtration market size was valued at USD 5.13 billion in 2025 and is expected to be worth around USD 12.45 billion by 2035, exhibiting a compound annual growth rate (CAGR) of 9.3% over the forecast period from 2026 to 2035. The pharmaceutical membrane filtration market is primarily driven by the rapid global expansion of biopharmaceutical production, which increasingly relies on advanced filtration technologies to ensure product safety and quality. The shift from chemical synthesis to biological expression systems requires specific filtration processes to protect the structural integrity of sensitive proteins and vaccines. Biological agents are more susceptible to thermal and chemical degradation than small molecules and are often validated using membrane filtration. Additionally, the growing burden of chronic diseases will continue to drive the large-scale production of monoclonal antibodies and recombinant proteins, all of which rely on high-throughput membrane systems.
Regulatory frameworks and the transition towards advanced manufacturing practices are also significantly contributing to market growth. Regulatory authorities such as the FDA and EMA mandate strict compliance with sterile filtration and viral clearance requirements, particularly in biologics and injectable drug production, where even minimal contamination is unacceptable. Furthermore, a “zero-contamination” threshold is no longer an aspiration but a requirement for market entry, forcing manufacturers to invest in validated filtration. At the same time, the industry is witnessing a shift towards continuous manufacturing and process intensification, where filtration is integrated directly into production workflows to enhance efficiency and reduce costs.
High adoption of technology in the pharmaceutical membrane filtration market
A significant trend in the pharmaceutical membrane filtration sector is the growing adoption of digital technologies and Industry 4.0 applications. Advanced technologies such as artificial intelligence (AI) and machine learning (ML) are being integrated into filtration processes to enhance membrane efficiency and deepen understanding of fouling behaviour. These tools enable manufacturers to analyse complex parameters such as water flux, predict micropollutant interactions, and optimise operating conditions in real time. As a result, filtration systems are becoming more intelligent, adaptive, and efficient. This shift towards "smart filtration" also enables predictive maintenance, reducing the risk of product batch failures caused by membrane breakage or excessive area fouling. In addition, the trend towards developing sustainable treatment strategies is growing as the industry faces pressure to limit environmental impact.
What is the construction of membranes used in pharmaceutical filtration?
Filtration membranes used in pharmaceutical applications are typically made of polymeric materials with highly controlled pore structures in the micron-to-submicron range. These membranes are generally thin, measuring about 120 micrometres, and require careful handling due to their delicate structure. Functionally, they act as selective barriers, trapping particulates and microorganisms on their surface while allowing the desired fluid to pass through, much like a sieve.
Membranes are available in a wide range of pore sizes, typically from approximately 0.010 ± 0.002 µm to 5.0 ± 1.2 µm, enabling their use across diverse filtration applications. For instance, smaller pore sizes (0.010–0.10 µm) are effective for removing viruses from air and water, while slightly larger pores (0.30–0.65 µm) are commonly used for removing bacteria. Larger-pore membranes are applied in specialised areas such as aerosol analysis, radioactivity testing, and particle sizing.
In terms of structural design, membrane filters are manufactured in different module configurations, including flat-sheet and hollow-fibre formats. During module construction, the membrane material is carefully integrated and sealed into assemblies to ensure mechanical stability and consistent performance. Hollow-fibre modules, in particular, offer high surface area and efficiency, making them suitable for large-scale applications. Additionally, spiral-wound modules are widely used for long-term operations due to their durability and ability to handle continuous filtration processes.
How to use a microfiltration membrane?
Cleaning: Wash the filter membrane with distilled water before use, then soak it in distilled water at 70-80℃ for 4 hours, or at room temperature for 12 hours.
Disinfection: Place the cleaned filter membrane in the filter and sterilize it by steam autoclaving at 120°C for 30 minutes. Other sterilization methods can also be used.
The filter membrane should be wet before filtering liquids; otherwise, filtration speed may decrease. If sterilised and dry, it must be wetted with sterile water before use.
Report Scope
| Area of Focus | Details |
| Market Size in 2026 | USD 5.61 Billion |
| Market Size in 2035 | USD 12.45 Billion |
| CAGR 2026 to 2035 | 9.30% |
| Dominant Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Key Segments | Products, Scale, Usage, Technique, Application, End-user, Region |
| Key Companies | Merck KGaA, Danaher Corporation, Sartorius AG, Thermo Fisher Scientific, Parker Hannifin Corporation, 3M Company, Eaton Corporation plc, Donaldson Company, Inc., Porvair plc, Alfa Laval, Corning Inc., Saint-Gobain, MANN+HUMMEL, Meissner Filtration Products, Repligen Corporation |
Business Expansion and Merger Strategies
Rapid business expansion and strategic mergers are significantly driving growth in the pharmaceutical membrane filtration market. In 2024, Sartorius AG announced a multi-million-dollar investment to expand its membrane production capabilities in France and Germany. This expansion is a direct response to a 20% increase in demand for polyethersulfone (PES) membranes in the post-pandemic environment. These developments reflect strong long-term confidence in market growth and underscore a strategy focused on securing the supply chain against future disruptions. By increasing domestic production capacity, these corporations are poised to better serve the European and North American markets by reducing lead times.
Novel Product Innovations in Sterile Filtration
Continuous innovation in sterile filtration technologies remains a major growth driver for pharmaceutical membrane filtration. In 2025, Merck KGaA introduced a cutting-edge ultra-low-binding membrane specifically for high-concentration protein therapies. This innovation addresses a critical challenge in bioprocessing, namely the loss of high-cost active pharmaceutical ingredients (APIs) due to non-specific adsorption to the filter membrane surface. In addition, the new membrane technology can save manufacturers millions of dollars annually by maximising product recovery. This innovation illustrates the industry's ongoing transition towards high-efficiency, application-specific filtration solutions.
Government Programs and Investment in Healthcare Infrastructure
Government initiatives and investments in healthcare infrastructure are playing a crucial role in shaping pharmaceutical membrane filtration. Programs such as the "Bio-Secure" initiatives launched in the United States and the European Union provide governments with substantial grants to produce membranes domestically. The purpose of these initiatives is to "reshore" pharmaceutical manufacturing and reduce dependence on global supply chains, particularly for critical healthcare components. For example, the U.S. government has committed funds to modernising domestic filtration manufacturing, recognising its importance for ensuring national health security. This type of government support will help to stabilise market growth and enable domestic technological development.
Strategic Partnerships for Global Supply Chain Reliability
Strategic partnerships are increasingly shaping the development and reliability of global supply chains in pharmaceutical membrane filtration. In 2025, Danaher Corporation announced plans to integrate AI-based sensors into its filtration systems. This marks a clear shift from reactive to proactive maintenance, enabling real-time prediction of membrane fouling. Strategic partnerships between filtration experts and software developers are becoming more common, reflecting the industry’s move towards smart manufacturing. These partnerships will be valuable in developing the "smart factories" of the future, where filtration processes are fully integrated and optimised within the broader production data set.
The pharmaceutical membrane filtration market is segmented by region into North America, Europe, Asia-Pacific, and LAMEA. Here is a brief overview of each region:
The Asia-Pacific pharmaceutical membrane filtration market size was accounted for USD 1.23 billion in 2025 and is projected to hit USD 2.99 billion by 2035. The Asia Pacific region is the fastest-growing market, driven by significant investment in biosimilar production and the creation of local vaccine manufacturing hubs. Governments across the region are actively supporting the development of bioprocessing infrastructure to reduce their dependence on Western countries for drug products. In addition, leading Contract Development and Manufacturing Organizations (CDMOs) are expanding their "single-use" footprint in Singapore and South Korea to meet global demand for biologics. National initiatives such as "China 2025" and "Make in India" are supported by the adoption of advanced medical technologies.
China: Scale Up Infrastructure and Leadership in Biopharmaceuticals
China is rapidly transitioning from a manufacturing-focused economy to a global leader in biopharmaceutical innovation, supported by substantial government investments in dedicated biotechnology parks.
India: The World's Population of Apothecaries
India continues to hold its position as the “pharmacy of the world,” with its pharmaceutical membrane filtration market driven by the large-scale production and export of generic drugs and biosimilars.
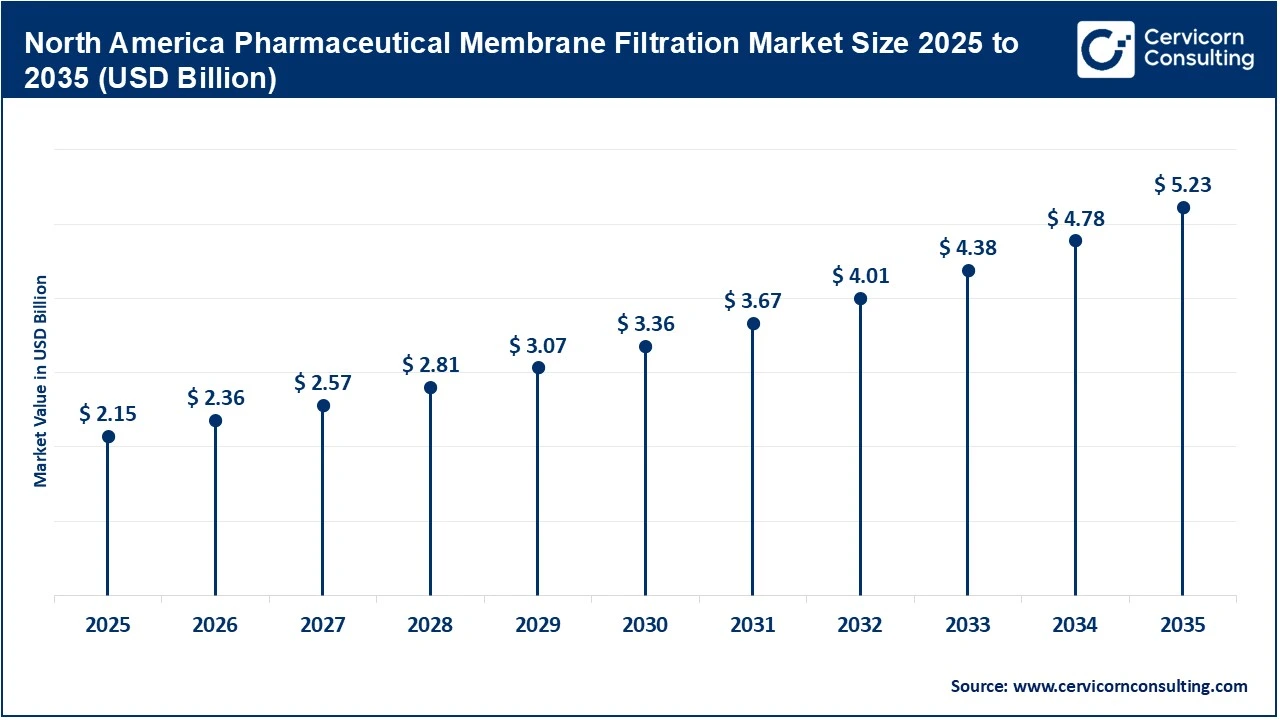
The North America pharmaceutical membrane filtration market size was reached at USD 2.15 billion in 2025 and is forecasted to garner around USD 5.23 billion by 2035.
North America continues to hold the largest share, supported by its leadership in research and development, particularly in cell and gene therapies (CGT). These advanced therapies require small-scale, specialised filtration systems with extreme precision, which command a higher price. The region is also characterised by high adoption of automated integrity testing and digital twin technology, which enhance process efficiency and reliability. Furthermore, regulatory initiatives, including the U.S. FDA’s focus on advanced manufacturing, are encouraging pharmaceutical companies to transition from traditional batch processes to continuous filtration workflows to improve drug quality and enhance supply chain resilience.
United States: Market Maturity and High-Value R&D Focus
The United States market is widely regarded as the global benchmark for pharmaceutical filtration, largely because of the high concentration of biotech startups and multinational pharmaceutical companies located there.
Canada: Growth in Specialized Bioprocessing Facilities
Canada is well-established in specialized vaccine production and biologics R&D in the Ontario and Quebec life sciences corridors.
The Europe pharmaceutical membrane filtration market size was estimated at USD 1.39 billion in 2025 and is projected to surpass around USD 3.36 billion by 2035. Europe is known for the most rigorous regulatory standards globally and a distinct focus on environmental sustainability. Regulatory initiatives, such as those of the European Medicines Agency (EMA), have set high standards for viral clearance, and high-flux nanofiltration is required for biologics. Additionally, Europe is at the forefront of the "green membrane" movement, with companies such as Merck and Sartorius investing in solvent-free membrane technologies. The European Union’s "Green Deal" has motivated pharmaceutical companies in the region to audit the lifecycle of their single-use plastic waste, prompting them to issue guidelines for new circular-economy solutions for membrane disposal in the coming year.
United Kingdom: Strategic Pivot toward Advanced Life Sciences
The United Kingdom is increasingly positioning itself as a global hub for advanced life sciences, with a strong focus on high-value biologics and genomics.
Germany: Engineering Dominance in Pharmaceutical Equipment
Germany continues to lead the global market for high-end pharmaceutical machinery and membrane modules, supported by its reputation for precision engineering and product reliability.
Pharmaceutical Membrane Filtration Market Share, By Region, 2025 (%)
| Region | Revenue Share, 2025 (%) |
| North America | 42% |
| Europe | 27% |
| Asia Pacific | 24% |
| LAMEA | 7% |
The LAMEA pharmaceutical membrane filtration market was valued at USD 0.36 billion in 2025 and is anticipated to reach around USD 0.87 billion by 2035. The LAMEA region is undergoing a significant shift towards strengthening domestic pharmaceutical manufacturing capabilities to improve healthcare security and accessibility. The Middle East, particularly the United Arab Emirates (UAE) and Saudi Arabia, has decided to invest in high-tech sectors of the healthcare industry to diversify their economies beyond oil. Meanwhile, in Latin America, countries like Brazil are promoting vaccine production through public-private partnerships, supporting the growth of the pharmaceutical sector. Across the region, there is high adoption of basic membrane filtration units to develop the generic drug manufacturing capability of their nations, which is increasing demand to modernize their healthcare infrastructure and improve access to affordable essential medicines.
Brazil: Expansion of Domestic Vaccine and Biologic Production
Brazil is focused on reducing dependence on the trade balance in pharmaceuticals, especially high-cost biologics produced through public-private partnerships.
United Arab Emirates: Diversification into High-Tech Healthcare Areas
The United Arab Emirates is emerging as a life sciences hub for the Middle East, with several investments in next-generation manufacturing facilities across the Global South.
The pharmaceutical membrane filtration market is segmented into products, scale, usage, technique, application, end-user, and region.
Membrane filters are the leading product category in the pharmaceutical filtration market, primarily because they are high-velocity consumables. In the pharmaceutical processing industry, membrane filters are used throughout the process, from initial buffer preparation to final sterile filtration, to remove particles, microorganisms, and other contaminants. The significance of these filters ties to the "razor-and-blade" business model, where the need to replace filters frequently drives increases in consumables revenue and process repeatability.
Pharmaceutical Membrane Filtration Market Share, By Products, 2025 (%)
| Products | Revenue Share, 2025 (%) |
| Membrane Filters | 52% |
| Filtration Systems | 33% |
| Consumables & Others | 15% |
Filtration systems are the fastest-growing product segment in the pharmaceutical filtration market, mainly driven by the industry’s shift towards automation and Industry 4.0 practices. Modern biopharmaceutical manufacturing increasingly relies on integrated systems that go beyond basic filtration, incorporating real-time monitoring of parameters such as pressure, flow rates, and filter integrity. These advanced systems, often aligned with sensors, control software, and automated components, help reduce manual intervention, minimize human error, and improve process consistency.
The commercial-scale segment remains the largest operating segment in the pharmaceutical membrane filtration market, as it represents the final stage of drug manufacturing and has the highest production volumes. These processes handle thousands of litres of media, buffers, and active pharmaceutical ingredients (APIs) used to treat patients worldwide. Additionally, commercial manufacturing is typically governed by long-term regulatory approvals and contracts, which “lock in” specific filtration components and technologies.
Pharmaceutical Membrane Filtration Market Share, By Scale, 2025 (%)
| Scale | Revenue Share, 2025 (%) |
| Commercial Scale | 64% |
| Pilot-scale | 21% |
| R&D Scale | 15% |
R&D-scale filtration is the fastest-growing segment of the market, primarily due to the rapid expansion of early-stage drug discovery and the growth of advanced, specialised therapeutic modalities. Areas such as personalised medicine, orphan drugs, and cell and gene therapies have required the industry to move towards smaller, high-value batches that need precise benchtop filtration. In R&D and clinical trial settings, researchers require flexible filtration tools that can accommodate a wide range of experimental conditions with minimal product loss, especially when drug candidates are extremely costly or in short supply.
Single-use (SUT) systems are becoming the dominant usage pattern in the market, mainly due to their operational flexibility and reduced risk of contamination. In conventional stainless-steel manufacturing operations, extensive cleaning-in-place (CIP) and sterilisation-in-place (SIP) procedures are required. These processes are time-consuming, water-intensive, and carry a potential risk of cross-contamination. On the other hand, single-use technologies are widely adopted, particularly in modern biopharmaceutical facilities.
Pharmaceutical Membrane Filtration Market Share, By Usage, 2025 (%)
| Usage | Revenue Share, 2025 (%) |
| Single Use | 68% |
| Reusable | 32% |
Reusable filtration solutions are among the fastest-growing sectors in the market, primarily driven by increasing economic and environmental considerations. The widespread adoption of single-use systems has raised concerns about plastic waste and long-term costs, especially in large-scale manufacturing operations. For high-demand products such as vaccines and monoclonal antibodies, the cumulative expense of disposable components can become significant. As a result, manufacturers are revisiting reusable systems, which, despite higher initial investment, offer cost efficiency and sustainability over time.
Microfiltration is the leading filtration technique in the market, serving as the fundamental process for particle removal and microbial control. Generally, microfiltration is used to separate suspended solids, bacteria, and yeast from process streams, making it an essential step in applications such as cell harvesting, clarification, and sterile filtration. The dominance of microfiltration stems from its universal use in the manufacture of small- and large-molecule pharmaceuticals. Moreover, liquid pharmaceutical products are processed through a microfiltration membrane during manufacture to meet requirements for clarity and safety.
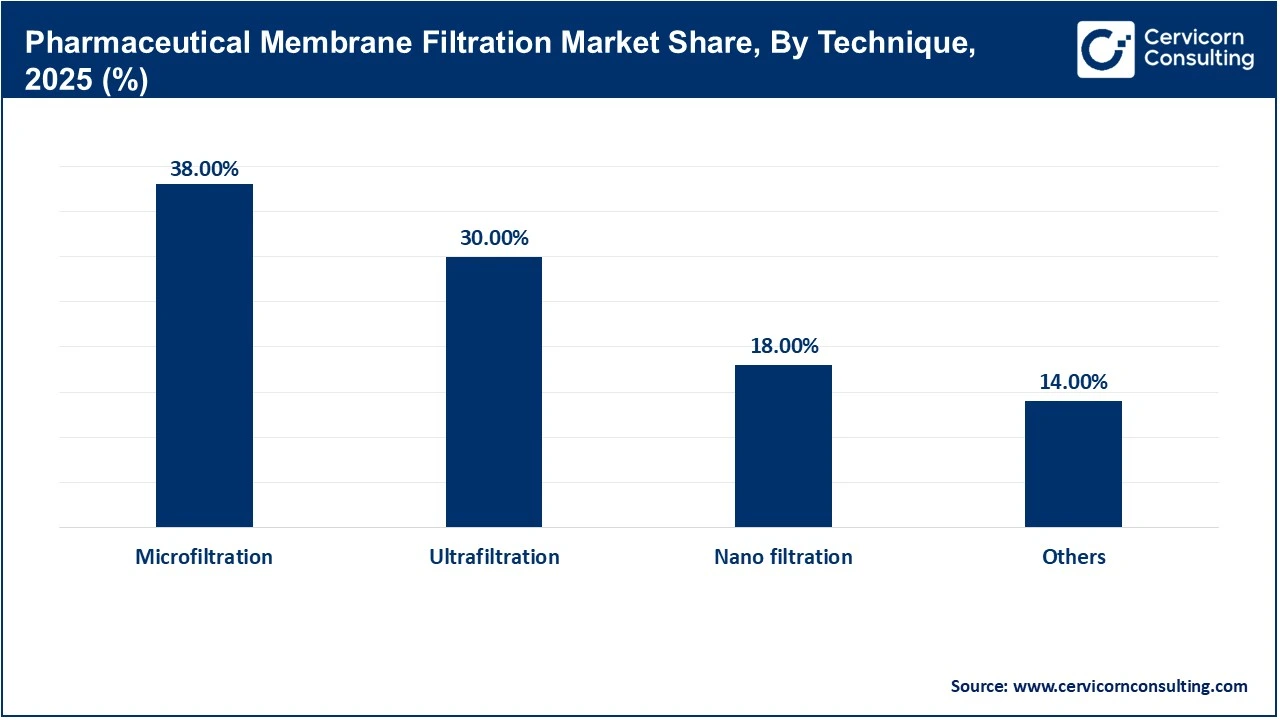
Nanofiltration is the fastest-growing filtration technique, driven by the increasing complexity of modern biopharmaceutical products and the need to achieve separations on a molecular scale. In contrast to microfiltration, which focuses on removing larger particles, nanofiltration is used to remove small organic molecules, viruses, and multivalent ions. This capability is particularly important in the purification of advanced biologics, where maintaining product integrity while ensuring safety is critical. As a result, demand for nanofiltration technologies is rising rapidly.
Final product processing remains the dominant application area for membrane filtration, as it is the most critical stage in the manufacturing life cycle, where the drug substance is prepared for patient administration and requires the highest levels of purity and sterility. Therefore, pharmaceutical companies typically invest in higher-cost, validated, premium-grade membrane filters during the final fluid processing step in drug manufacture to ensure product safety.
Pharmaceutical Membrane Filtration Market Share, By Application, 2025 (%)
| Application | Revenue Share, 2025 (%) |
| Final Product Processing | 40% |
| Raw Material Filtration | 20% |
| Virus Filtration | 18% |
| Cell Separation | 12% |
| Others | 10% |
Virus filtration is the fastest-growing application in the market, supported by tightening global regulatory standards and the risk associated with mammalian cell products. Regulatory agencies, such as the FDA and EMA, have established strict guidelines that require multiple viral clearance steps to ensure product safety. Virus-retentive membranes are designed to remove extremely small viral particles while allowing the target protein to pass through, which typically requires outstanding membrane consistency and integrity.
Pharmaceutical and biotechnology companies are the largest end-user category in the market, mainly because they own most of the global drug pipelines and possess the manufacturing capacity to support global distribution. These organisations invest in large-scale filtration and high-volume membrane consumables to support commercial production. They remain dominant by consistently producing and providing full-service support for long-established manufacturing lines. Their end-to-end involvement in drug development, from discovery to commercialisation, means their internal purchasing decisions and technology preferences drive the overall direction and volume of the filtration market.
Pharmaceutical Membrane Filtration Market Share, By End-user, 2025 (%)
| End-user | Revenue Share, 2025 (%) |
| Pharmaceutical & Biotechnology Companies | 62% |
| CROs & CDMOs | 20% |
| Academic & Research Institutes | 12% |
| Others | 6% |
Contract Research Organisations (CROs) and Contract Development and Manufacturing Organisations (CDMOs) are the fastest-growing end-user category in the market, primarily due to a fundamental shift in pharmaceutical development and production. CDMOs are investing heavily in newer filtration technologies to offer contract manufacturing services to multiple clients that need flexibility and efficiency to convert higher volumes of product simultaneously. In addition, many small and mid-size biotech companies do not have their own manufacturing facilities. Therefore, they use CDMOs exclusively for production, driving high demand for filtration solutions resulting from outsourcing.
By Products
By Scale
By Usage
By Technique
By Application
By End-user
By Region
Chapter 1. Market Introduction and Overview
1.1 Market Definition and Scope
1.1.1 Overview of Pharmaceutical Membrane Filtration
1.1.2 Scope of the Study
1.1.3 Research Timeframe
1.2 Research Methodology and Approach
1.2.1 Methodology Overview
1.2.2 Data Sources and Validation
1.2.3 Key Assumptions and Limitations
Chapter 2. Executive Summary
2.1 Market Highlights and Snapshot
2.2 Key Insights by Segments
2.2.1 By Products Overview
2.2.2 By Scale Overview
2.2.3 By Usage Overview
2.2.4 By Technique Overview
2.2.5 By End-user Overview
2.2.6 By Application Overview
2.3 Competitive Overview
Chapter 3. Global Impact Analysis
3.1 Russia-Ukraine Conflict: Global Market Implications
3.2 Regulatory and Policy Changes Impacting Global Markets
Chapter 4. Market Dynamics and Trends
4.1 Market Dynamics
4.1.1 Market Drivers
4.1.2 Market Restraints
4.1.3 Market Opportunities
4.1.4 Market Challenges
4.2 Market Trends
Chapter 5. Premium Insights and Analysis
5.1 Global Pharmaceutical Membrane Filtration Market Dynamics, Impact Analysis
5.2 Porter’s Five Forces Analysis
5.2.1 Bargaining Power of Suppliers
5.2.2 Bargaining Power of Buyers
5.2.3 Threat of Substitute Products
5.2.4 Rivalry among Existing Firms
5.2.5 Threat of New Entrants
5.3 PESTEL Analysis
5.4 Value Chain Analysis
5.5 Product Pricing Analysis
5.6 Vendor Landscape
5.6.1 List of Buyers
5.6.2 List of Suppliers
Chapter 6. Pharmaceutical Membrane Filtration Market, By Products
6.1 Global Pharmaceutical Membrane Filtration Market Snapshot, By Products
6.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2035
6.1.1.1 Membrane Filters
6.1.1.2 Filtration Systems
6.1.1.3 Consumables & Others
Chapter 7. Pharmaceutical Membrane Filtration Market, By Scale
7.1 Global Pharmaceutical Membrane Filtration Market Snapshot, By Scale
7.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2035
7.1.1.1 Commercial Scale
7.1.1.2 Pilot-scale
7.1.1.3 R&D Scale
Chapter 8. Pharmaceutical Membrane Filtration Market, By Usage
8.1 Global Pharmaceutical Membrane Filtration Market Snapshot, By Usage
8.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2035
8.1.1.1 Single Use
8.1.1.2 Reusable
Chapter 9. Pharmaceutical Membrane Filtration Market, By Technique
9.1 Global Pharmaceutical Membrane Filtration Market Snapshot, By Technique
9.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2035
9.1.1.1 Microfiltration
9.1.1.2 Ultrafiltration
9.1.1.3 Nano filtration
9.1.1.4 Others
Chapter 10. Pharmaceutical Membrane Filtration Market, By End-user
10.1 Global Pharmaceutical Membrane Filtration Market Snapshot, By End-user
10.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2035
10.1.1.1 Pharmaceutical & BioTechnique Companies
10.1.1.2 CROs & CDMOs
10.1.1.3 Academic & Research Institutes
10.1.1.4 Others
Chapter 11. Pharmaceutical Membrane Filtration Market, By Application
11.1 Global Pharmaceutical Membrane Filtration Market Snapshot, By Application
11.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2035
11.1.1.1 Final Product Processing
11.1.1.2 Raw Material Filtration
11.1.1.3 Virus Filtration
11.1.1.4 Cell Separation
11.1.1.5 Others
Chapter 12. Pharmaceutical Membrane Filtration Market, By Region
12.1 Overview
12.2 Pharmaceutical Membrane Filtration Market Revenue Share, By Region 2024 (%)
12.3 Global Pharmaceutical Membrane Filtration Market, By Region
12.3.1 Market Size and Forecast
12.4 North America
12.4.1 North America Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.4.2 Market Size and Forecast
12.4.3 North America Pharmaceutical Membrane Filtration Market, By Country
12.4.4 U.S.
12.4.4.1 U.S. Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.4.4.2 Market Size and Forecast
12.4.4.3 U.S. Market Segmental Analysis
12.4.5 Canada
12.4.5.1 Canada Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.4.5.2 Market Size and Forecast
12.4.5.3 Canada Market Segmental Analysis
12.4.6 Mexico
12.4.6.1 Mexico Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.4.6.2 Market Size and Forecast
12.4.6.3 Mexico Market Segmental Analysis
12.5 Europe
12.5.1 Europe Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.5.2 Market Size and Forecast
12.5.3 Europe Pharmaceutical Membrane Filtration Market, By Country
12.5.4 UK
12.5.4.1 UK Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.5.4.2 Market Size and Forecast
12.5.4.3 UK Market Segmental Analysis
12.5.5 France
12.5.5.1 France Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.5.5.2 Market Size and Forecast
12.5.5.3 France Market Segmental Analysis
12.5.6 Germany
12.5.6.1 Germany Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.5.6.2 Market Size and Forecast
12.5.6.3 Germany Market Segmental Analysis
12.5.7 Rest of Europe
12.5.7.1 Rest of Europe Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.5.7.2 Market Size and Forecast
12.5.7.3 Rest of Europe Market Segmental Analysis
12.6 Asia Pacific
12.6.1 Asia Pacific Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.6.2 Market Size and Forecast
12.6.3 Asia Pacific Pharmaceutical Membrane Filtration Market, By Country
12.6.4 China
12.6.4.1 China Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.6.4.2 Market Size and Forecast
12.6.4.3 China Market Segmental Analysis
12.6.5 Japan
12.6.5.1 Japan Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.6.5.2 Market Size and Forecast
12.6.5.3 Japan Market Segmental Analysis
12.6.6 India
12.6.6.1 India Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.6.6.2 Market Size and Forecast
12.6.6.3 India Market Segmental Analysis
12.6.7 Australia
12.6.7.1 Australia Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.6.7.2 Market Size and Forecast
12.6.7.3 Australia Market Segmental Analysis
12.6.8 Rest of Asia Pacific
12.6.8.1 Rest of Asia Pacific Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.6.8.2 Market Size and Forecast
12.6.8.3 Rest of Asia Pacific Market Segmental Analysis
12.7 LAMEA
12.7.1 LAMEA Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.7.2 Market Size and Forecast
12.7.3 LAMEA Pharmaceutical Membrane Filtration Market, By Country
12.7.4 GCC
12.7.4.1 GCC Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.7.4.2 Market Size and Forecast
12.7.4.3 GCC Market Segmental Analysis
12.7.5 Africa
12.7.5.1 Africa Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.7.5.2 Market Size and Forecast
12.7.5.3 Africa Market Segmental Analysis
12.7.6 Brazil
12.7.6.1 Brazil Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.7.6.2 Market Size and Forecast
12.7.6.3 Brazil Market Segmental Analysis
12.7.7 Rest of LAMEA
12.7.7.1 Rest of LAMEA Pharmaceutical Membrane Filtration Market Revenue, 2022-2035 ($Billion)
12.7.7.2 Market Size and Forecast
12.7.7.3 Rest of LAMEA Market Segmental Analysis
Chapter 13. Competitive Landscape
13.1 Competitor Strategic Analysis
13.1.1 Top Player Positioning/Market Share Analysis
13.1.2 Top Winning Strategies, By Company, 2022-2024
13.1.3 Competitive Analysis By Revenue, 2022-2024
13.2 Recent Developments by the Market Contributors (2024)
Chapter 14. Company Profiles
14.1 Merck KGaA
14.1.1 Company Snapshot
14.1.2 Company and Business Overview
14.1.3 Financial KPIs
14.1.4 Product/Service Portfolio
14.1.5 Strategic Growth
14.1.6 Global Footprints
14.1.7 Recent Development
14.1.8 SWOT Analysis
14.2 Danaher Corporation
14.3 Sartorius AG
14.4 Thermo Fisher Scientific
14.5 Parker Hannifin Corporation
14.6 3M Company
14.7 Eaton Corporation plc
14.8 Donaldson Company, Inc.
14.9 Porvair plc
14.10 Alfa Laval
14.11 Corning Inc.
14.12 Saint-Gobain
14.13 MANN+HUMMEL
14.14 Meissner Filtration Products
14.15 Repligen Corporation