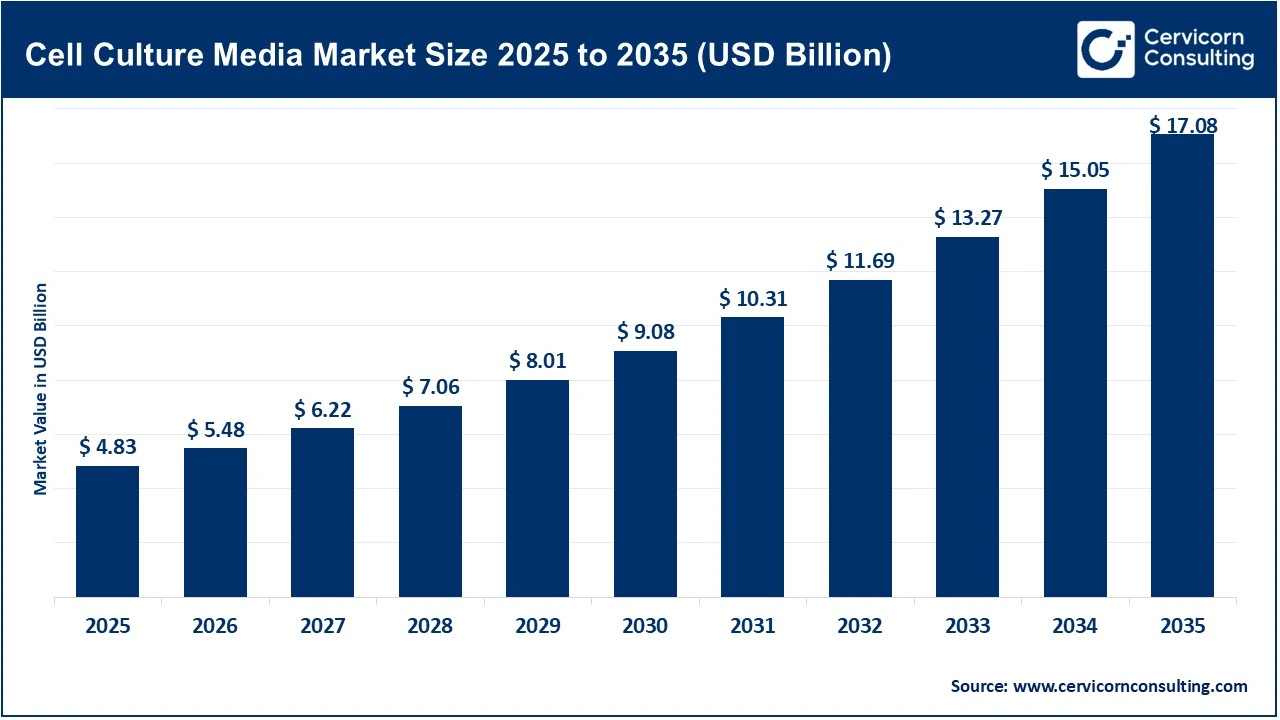
The global cell culture media market size reached at USD 4.83 billion in 2025 and is expected to be worth around USD 17.08 billion by 2035, exhibiting a compound annual growth rate (CAGR) of 13.5% over the forecast period from 2026 to 2035. The cell culture media market is experiencing significant growth, mainly driven by the rapid expansion of biopharmaceutical research and development (R&D) activities. Global investment in biopharma R&D exceeds USD 200 billion annually, with a large share of these funds allocated to developing advanced biologics such as monoclonal antibodies (mAbs), recombinant proteins, and viral vectors. Additionally, the biologic pipeline is expanding and now accounts for nearly 40% of all drugs in development, directly increasing demand for higher-performance cell culture media. The production of mAbs will remain the largest consumer of cell culture media, with large-scale manufacturing facilities using thousands of liters of specialized media to sustain high cell densities and protein titers. Advanced media formulations can boost protein yields by up to 30%, which directly impacts the viability of biotech production.
In biopharmaceutical progress, the rising global prevalence of chronic diseases such as cancer, diabetes, and cardiovascular conditions is another key driver of market growth. For example, cancer incidence is projected to rise by nearly 50% by 2040, further increasing the demand for effective treatment options. This growing disease burden is also shifting focus toward targeted therapy and cell-based immunotherapy, such as CAR-T cell therapy. Moreover, the emergence of personalized medicine is fueling demand for primary cell culture and patient-specific disease modeling. The concept of chronic disease management has created a need for continuous production of recombinant proteins, such as insulin and erythropoietin, which are produced in cell culture systems.
Commonly Used Cell Lines, Culture Media, and Applications
This table highlights commonly used cell lines, including their morphology, species of origin, culture media requirements, and key applications. It aims to help readers understand how cell culture media are practically applied in various research and biopharmaceutical contexts, demonstrating how media composition varies depending on cell type and experimental objectives.
| Cell Line | Morphology | Species | Medium Composition | Applications |
| HeLa B | Epithelial | Human | MEM + 2mM Glutamine + 10% FBS + 1% Non-Essential Amino Acids (NEAA) | Tumourigenicity and virus studies |
| HL60 | Lymphoblast | Human | RPMI 1640 + 2mM Glutamine + 10–20% FBS | Differentiation studies |
| 3T3 clone A31 | Fibroblast | Mouse | DMEM + 2mM Glutamine + 5% New Born Calf Serum (NBCS) + 5% FBS | Tumourigenicity and virus studies |
| COS-7 | Fibroblast | Monkey | DMEM + 2mM Glutamine + 10% FBS | Gene expression and virus replication studies |
| CHO | Epithelial | Hamster | Ham’s F12 + 2mM Glutamine + 10% FBS | Nutritional and gene expression studies |
| HEK 293 | Epithelial | Human | EMEM (EBSS) + 2mM Glutamine + 1% Non-Essential Amino Acids (NEAA) + 10% FBS | Transformation studies |
| HUVEC | Epithelial | Human | F-12K + 10% FBS + 100 µg/ml Heparin | Angiogenesis studies |
| Jurkat | Lymphoblast | Human | RPMI-1640 + 10% FBS | Signaling studies |
Integration of High-Throughput Screening and Bioprocess Automation Driving the Market Growth
The integration of automation and high-throughput screening (HTS) is a major driver of market growth, significantly enhancing media optimization. Finding the right mix of nutrients for a specific cell line was once a tedious "trial and error" process, where different combinations had to be tested one at a time. Today, researchers utilise automated bioprocess development platforms to evaluate thousands of media variations simultaneously. Advanced platform technologies for bioprocess development maintain consistency across unit operations and scales, simplifying the planning and execution of experiments. These systems also facilitate the identification of the best lysis buffer compositions and product formation profiles by using parallel fed-batch cultures at the milliliter scale.
What are the major challenges for expanding cell culture media from laboratory research to industrial scale?
One of the major challenges in scaling cell culture media from laboratory research to industrial scale is maintaining nutritional uniformity and gas exchange efficiency across larger volumes. As bioprocesses move from small-scale systems like 1L shake flasks to large-scale 2,000L stirred-tank bioreactors, the operating environment experiences significant physical stresses on the cells. Additionally, large-scale operations face issues related to uneven nutrient depletion rates, which can cause inconsistent culture performance if not properly managed. To tackle these challenges, about 90% of large-scale facilities are shifting to dry powder media (DPM) formats, which are favoured for large-scale applications because they cut shipping costs by 80%, offer longer shelf life, and can be hydrated onsite with automated media preparation systems that ensure the media remains fresh and consistent for high-density cultures.
1. Corporate Expansion and Strategic Global Moves
Corporate expansion drives growth in the cell culture market. For example, Thermo Fisher Scientific announced a USD 100 million investment to expand its dry powder media manufacturing in the United States. This move was purely strategic to address supply chain vulnerabilities from previous years and the rising demand for local, high-volume manufacturing. Similarly, Sartorius AG has adopted an aggressive acquisition strategy of specialized media suppliers to enhance its "Total Solution" media portfolio, while now focusing particularly on the high-growth cell and gene therapy (CGT) sectors. Additionally, companies have reduced lead times by over 40% for biopharma clients when producing media locally.
2. Government Initiatives and Research Funding
Government initiatives and research funding have played a major role in the cell culture market. In the European Union, the Horizon Europe program has allocated over USD 50 million specifically for the development of sustainable and innovative cell-culture media. This includes funding for emerging areas such as "clean meat" media and advanced therapies. In countries like the UK and Ireland, biofabrication is actively promoted and focused on healthcare innovation, such as drug delivery, in vitro models, organoids, and tissue engineering. This commitment to developing ethically compliant healthcare solutions also supports environmentally friendly systems.
3 Advancements in Manufacturing Infrastructure
Technological advancements in manufacturing infrastructure represent a significant milestone in the cell culture market. In 2024, the industry is increasingly adopting fully automated or “lights-out” media preparation facilities. These facilities utilize robotic technology along with advanced sensors to mix, sterilize, and package media with minimal human interaction. Such industry requirements for extreme precision, where even a 1% variation in the amount of a trace element can impact the altered glycosylation pattern of a therapeutic protein, are crucial. Meanwhile, automated preparation reduces batch failure rates by 25%, providing a significant economic advantage for large-scale biologics manufacturers.
4. Corporate Collaborations for Specialized Media for Rare Disease
Collaborations between media suppliers and biotech startups for Rare Disease are another major milestone in the market. These partnerships aim to address the unique challenges associated with culturing rare and patient-specific cell types. In 2023, a major collaboration was announced, which includes Merck KGaA and several academic institutions, to develop niche media for rare genetic disorders. As a result, these collaborations have led to specialized media developed with "tailored kits," which involve specific cytokine cocktails that were optimized to account for the low-abundance cell types. This approach provides the potential for facilitating the clinical transition of rare disease therapies, which would originally take much longer.
The cell culture media market is segmented by region into North America, Europe, Asia-Pacific, and LAMEA. Here is a brief overview of each region:
The Asia-Pacific cell culture media market size reached at USD 1.11 billion in 2025 and is expected to be worth around USD 3.93 billion by 2035. Asia-Pacific is the fastest-growing region in the cell culture media market, driven by a significant shift towards outsourcing bioprocessing activities and the rapid growth of healthcare infrastructure. International pharmaceutical companies are increasingly relocating manufacturing and research activities to countries like China, India, and South Korea to cut operational costs and take advantage of better Contract Development and Manufacturing Organization (CDMO) capabilities. As a result, the "outsourcing wave" is shifting from small laboratory media to large bulk media powders and liquid concentrates to support large-scale bioreactor operations.
China: Rapid Growth in Large-scale Biologics Production Facilities
China is the fastest-growing country and is becoming a global biomanufacturing hub, mainly driven by significant state investment and a growing domestic CDMO sector.
India: Dominance in Biosimilars and Cost-effective Media Formulations
India has large-scale production capabilities and is a centre for affordable biosimilars that need a lot of high-yield media.
Japan: Leading the World in Stem Cell Research and Regenerative Medicine Applications
Japan is a global leader in induced pluripotent stem cell (iPSC) research and in a regulatory framework for proof-of-concept in regenerative medicine.
The North America cell culture media market size was estimated at USD 1.93 billion in 2025 and is forecasted to surpass around USD 6.83 billion by 2035.
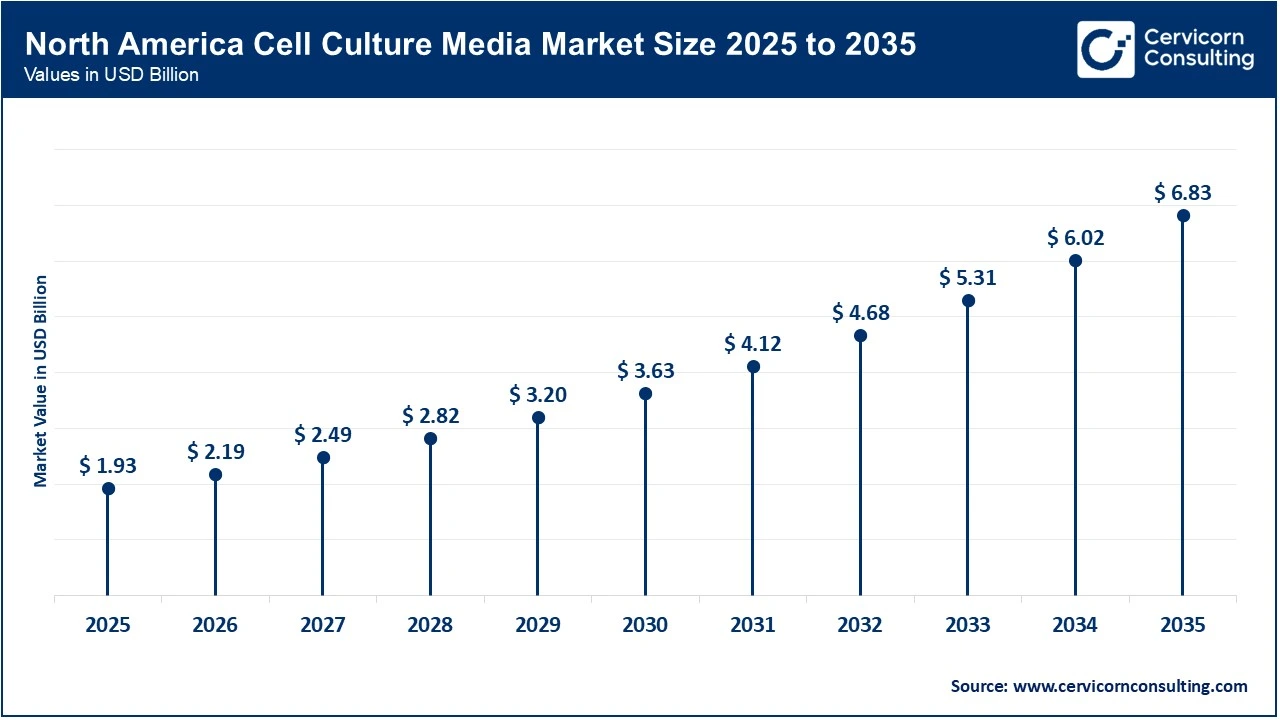
North America holds the largest share of the cell culture media market, mainly because of its high level of biopharmaceutical R&D activities. In the United States, biopharma companies spent over USD 100 billion on R&D this year, much of which focused on early-stage clinical trials for cell and gene therapies. This significant funding continues to boost demand for advanced research-grade cell culture media, including specialized formulations in the growing regenerative medicine market. Furthermore, North America remains a key market due to the more developed ecosystem among academic institutions and the availability of venture capital for many biotech start-ups.
United States: Continued Dominance from R&D and Clinical Development
The United States has upheld its role as the global leader in life sciences, high R&D investment, and advanced therapy development.
The Europe cell culture media market size was accounted for USD 1.30 billion in 2025 and is projected to hit around USD 4.61 billion by 2035. Europe’s market growth is primarily fueled by its strong position in the global biosimilars segment, supported by a favorable regulatory environment that promotes the development of cost-effective biologics. The region has approved over 90 biosimilars, establishing itself as the global leader in this field. European manufacturers are focusing on increasing efficiencies in bioprocessing to sustain current price levels for biosimilars. Consequently, the adoption of high-yield, chemically mediated processes has increased to reduce downstream processing complexities.
United Kingdom: Strategic Focus on Commercialization of Cell and Gene Therapy
The United Kingdom is positioning itself as a worldwide link between academic excellence and commercial manufacturing, particularly in the field of advanced therapy medicinal products (ATMPS).
Germany: European Hub for Pharmaceutical Manufacturing and Precision Engineering
Germany is the leading country in Europe's biopharmaceutical industry, emphasising engineering for bioprocessing and quality.
Cell Culture Media Market Share, By Region, 2025 (%)
| Region | Revenue Share, 2025 (%) |
| North America | 40% |
| Europe | 27% |
| Asia Pacific | 23% |
| LAMEA | 10% |
The LAMEA cell culture media market was valued at USD 0.48 billion in 2025 and is anticipated to reach around USD 1.71 billion by 2035. The LAMEA region currently accounts for a smaller share of the global market but is experiencing steady growth driven by increasing investments in local biopharmaceutical capabilities. Countries such as Brazil, South Africa, and the UAE are focused on establishing bioproduction hubs to reduce dependency on imports, and team trials are rising in the region due to the greater diversity of participants and comparatively lower drug development costs than other markets. Meanwhile, investments in Middle Eastern life science "Vision" programs are growing at approximately 20%. Furthermore, German companies are increasingly packaging media offerings with advanced bioreactor systems to deliver an integrated "plug-and-play" bioprocessing solution.
Brazil: Developing Local Bioproduction Capacity to Meet Regional Demand
Brazil is the largest market in South America for biopharmaceuticals, and it has been focusing on reducing its trade deficit by localising the manufacturing of biologics needed for care.
United Arab Emirates: Strategic Diversification into Life Sciences and Medical Hubs
The United Arab Emirates is using its sovereign capital to become a global hub for biotechnology by building top-tier life science clusters to diversify its economy.
The cell culture media market is segmented into type, product, application, end use, and region.
Liquid Media is a dominant type in the cell culture media market, mainly due to its natural usability, seamless integration into automated bioprocessing workflows, and its adoption as an industry standard for large-scale fermentation. Most industrial large-scale bioreactors are designed to operate in the liquid phase, enabling efficient nutrient delivery and waste removal. This makes liquid media essential for large-scale fermentation and biopharmaceutical manufacturing processes.
Cell Culture Media Market Share, By Type, 2025 (%)
| Type Segment | Revenue Share, 2025 (%) |
| Liquid Media | 55% |
| Semi-solid and Solid Media | 45% |
Semi-solid and solid media represent the fastest-growing segments in the market due to the rapid expansion of three-dimensional cell culture, oncology research, and colony-forming unit (CFU) assays. These media are vital for maintaining tissue architecture and enabling more physiologically relevant models for drug screening, especially in cancer research. Additionally, the industry is shifting toward greater physiological relevance and increased demand for specialized semi-solid matrices, such as those used in organoid culture.
Serum-Free Media is the leading product segment in the market because it is highly valued for regulatory compliance, batch consistency, and lower risk of viral contamination. By avoiding Fetal Bovine Serum, manufacturers can achieve more predictable cell growth and simplify downstream processing. As a result, this segment has become the standard method for producing therapeutic proteins and viral vectors, with over 80% of new biologic drug applications indicating the use of serum-free or chemically defined media.
Cell Culture Media Market Share, By Product, 2025 (%)
| Product Segment | Revenue Share, 2025 (%) |
| Serum-free Media | 32% |
| Classical Media | 16% |
| Stem Cell Culture Media | 14% |
| Chemically Defined Media | 12% |
| Specialty Media | 11% |
| Immunology Media | 10% |
| Other Cell Culture Media | 5% |
Stem Cell Culture Media is the fastest-growing segment in the market, mainly driven by the expansion of clinical trials in regenerative medicine and cell therapy. These cells require highly specific growth factors, including signaling molecules, to maintain pluripotency and direct differentiation into particular lineages. Additionally, more than 1,000 active cell and gene therapy trials are currently underway worldwide, leading to a surge in demand for high-performance stem cell media.
Biopharmaceutical production holds the largest market segment because it accounts for the highest volume of cell culture media consumption. Large-scale manufacturing of biologics, including monoclonal antibodies, insulin, and growth hormones, requires significant amounts of media, sometimes produced in 20,000-liter batches. This segment continues to lead due to the ongoing global demand for biologic drugs.
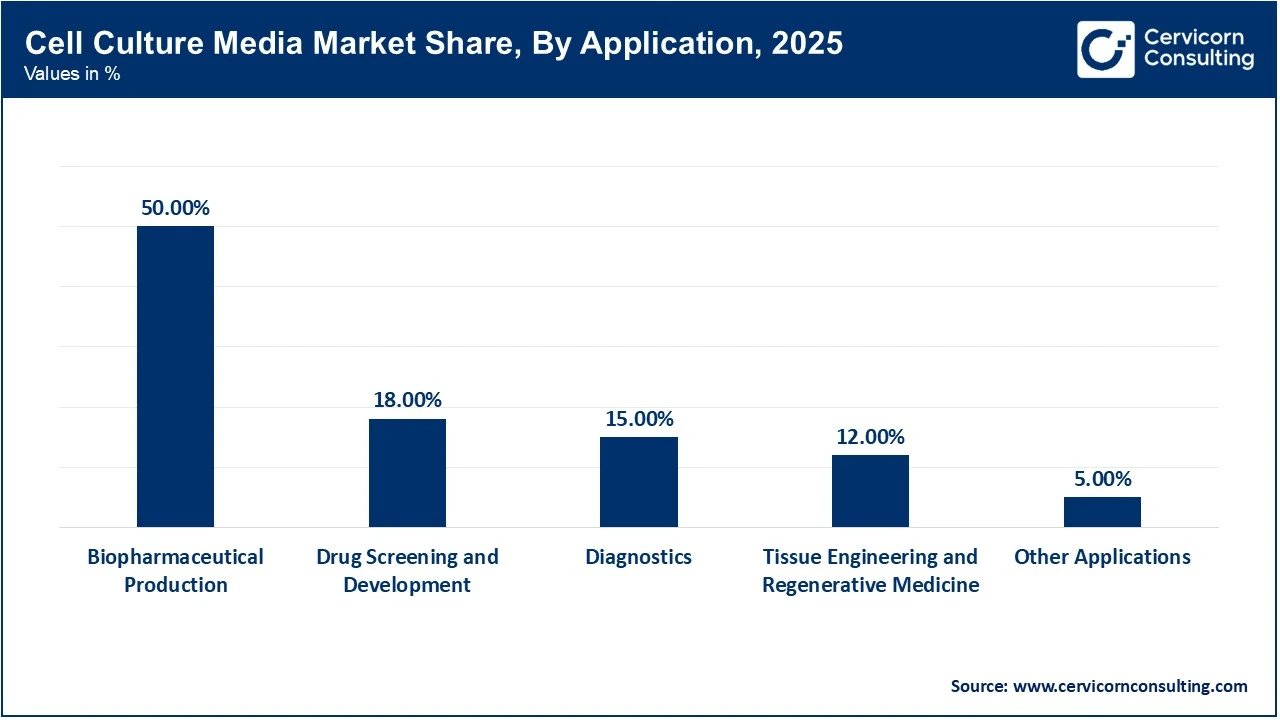
Tissue engineering and regenerative medicine is the fastest-growing segment, mainly because of the shift of tissue-engineered products from the lab to clinical applications. In advanced technologies like 3D bioprinting and directed assembly, there is a major focus on developing biomimetic scaffolds and organoids. As the FDA and EMA approve the first engineered tissues, such as' off-the-shelf' products for wound healing and organ repair, which require specialised media to support the complex metabolic needs of multi-cell type constructs, this segment is expected to grow exponentially.
Pharmaceutical and biotechnology companies lead the market, mainly supported by substantial investments in research and development and their role as primary producers of biologic drugs. These companies procure media at a large scale and frequently collaborate with media manufacturers to develop proprietary formulations. Their extensive use of media across drug discovery, development, and manufacturing reinforces their dominant market position.
Cell Culture Media Market Share, By End Use, 2025 (%)
| End Use Segment | Revenue Share, 2025 (%) |
| Pharmaceutical and Biotechnology Companies | 46% |
| Research and Academic Institutes | 24% |
| Hospitals and Diagnostic Laboratories | 20% |
| Other End Uses | 10% |
Hospitals and diagnostic laboratories are the fastest-growing end users in the market due to the decentralization of cell therapy and the increasing adoption of point-of-care diagnostics. As personalized therapies such as CAR-T become commonplace, many hospitals establish their own cell processing units to prepare therapies "at the bedside." Moreover, the use of cell-based assays for diagnostic purposes, such as testing patient-derived tumor cells against a range of chemotherapy agents, extends the panel of chemotherapies.
By Type
By Product
By Application
By End Use
By Region