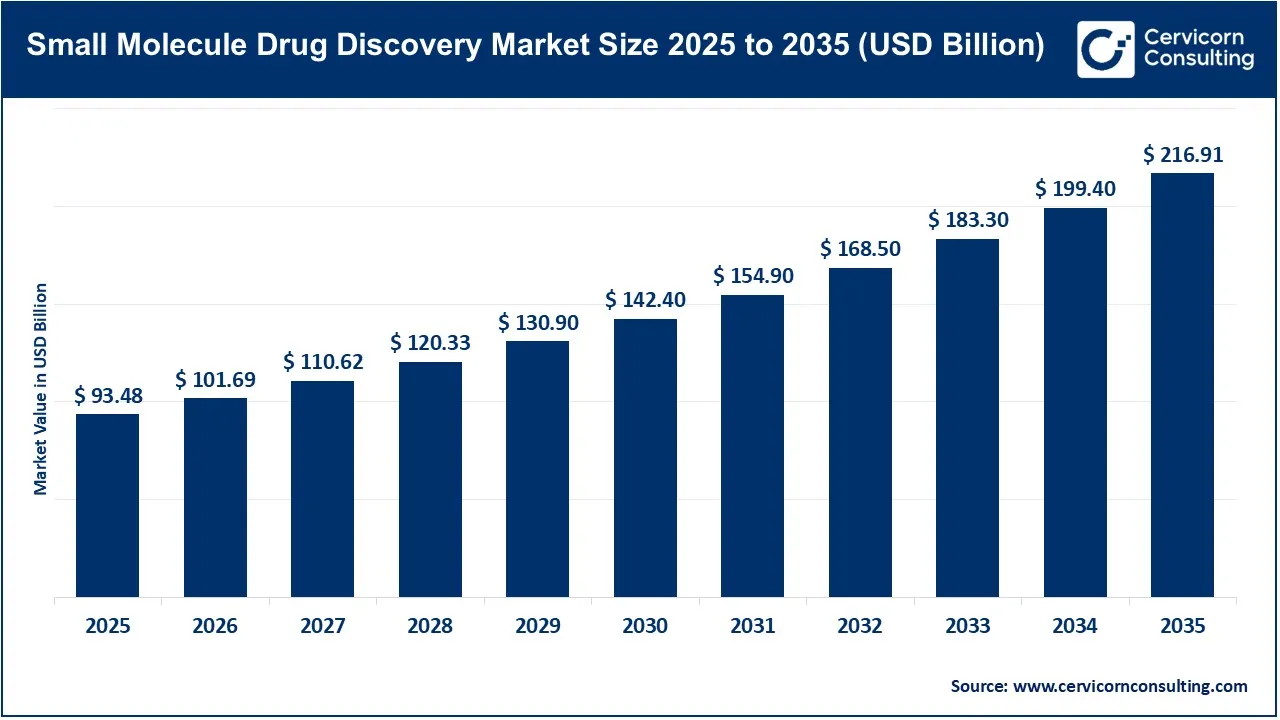
The global small molecule drug discovery market size was valued at USD 93.48 billion in 2025 and is expected to exceed around USD 216.91 billion by 2035, expanding at a compound annual growth rate (CAGR) of 8.8% over the forecast period from 2026 to 2035. The small molecule drug discovery market is primarily driven by the growing burden of chronic and complex diseases, particularly cancer, cardiovascular disorders, neurological diseases, and metabolic conditions, which continue to increase demand for targeted therapeutics. Small molecules remain highly preferred due to their oral bioavailability, lower manufacturing costs, and their ability to penetrate cell membranes effectively. Regulatory momentum is also supporting market expansion of the 55 new drugs approved by the U.S. FDA in 2023, approximately 62% were small-molecule drugs, demonstrating their continued dominance in pharmaceutical pipelines. In addition, the United States alone contributed more than 50% of the world’s new molecular discoveries over the past decade, reflecting strong innovation activity and sustained investment in drug research.
Another major growth factor in small molecule drug discovery market is the rapid adoption of AI, machine learning, and computational drug discovery platforms, which are significantly reducing discovery timelines and improving hit identification efficiency. Pharmaceutical companies are increasing R&D spending to accelerate molecule screening, lead optimization, and target validation; global pharma R&D expenditure exceeded USD 244 billion in 2023, with substantial investment directed toward small-molecule therapeutics and outsourcing partnerships with CROs. AI-enabled discovery platforms can now screen billions of virtual compounds, and some companies have reduced candidate nomination timelines from the traditional 5–6 years to as little as 18–24 months, greatly improving productivity. The growing use of high-throughput screening, fragment-based drug discovery, and precision medicine approaches is further driving market growth by enabling faster and more accurate development of novel therapies.
Rapid Progress in AI-Driven Hit-to-Lead Accelerating Small Molecule Drug Discovery Market Growth
The rapid advancement of AI-driven hit-to-lead (H2L) optimization is significantly accelerating the small molecule drug discovery market by reducing the time, cost, and failure rates associated with early-stage drug development. Traditionally, converting a biological “hit” into a viable lead compound required extensive medicinal chemistry cycles and years of experimental screening; however, AI-powered models now enable researchers to predict molecular binding, toxicity, pharmacokinetics, and compound optimization with much greater precision. This allows pharmaceutical and biotechnology companies to prioritize high-potential molecules earlier, reduce laboratory iterations, and improve success rates in candidate selection. AI also strengthens virtual screening capabilities by analyzing massive chemical libraries and identifying drug-like compounds that conventional methods may overlook. As a result, pharmaceutical companies are increasingly partnering with AI-driven discovery firms and integrating generative chemistry, predictive modeling, and automated screening into their R&D pipelines to improve productivity and shorten time-to-clinic, thereby driving demand across the small molecule drug discovery ecosystem.
Some of the key factors of this growth
Report Scope
| Area of Focus | Details |
| Market Size in 2026 | USD 101.69 Billion |
| Market Size in 2035 | USD 216.91 Billion |
| CAGR 2026 to 2035 | 8.80% |
| Dominant Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Key Segments | Drug Type, Drug Discovery Process/Workflow, Technology, Therapeutic Area, End User, Region |
| Key Companies | Pfizer, AstraZeneca, F. Hoffmann-La Roche (Roche), Novartis, Merck & Co., Bristol Myers Squibb, Johnson & Johnson (Janssen), Eli Lilly and Company, Thermo Fisher Scientific, Charles River Laboratories, Evotec, WuXi AppTec |
1. Takeda–Iambic AI Partnership for Small Molecule Design (2026)
In February 2026, Takeda Pharmaceutical entered a multi-year collaboration valued at more than USD 1.7 billion with AI biotech Iambic Therapeutics to design small-molecule therapies for oncology and gastrointestinal diseases using AI-driven molecular modeling. The partnership is important because it validates AI as a mainstream discovery engine rather than an experimental tool. By integrating Iambic’s NeuralPLexer platform to predict protein–molecule binding, Takeda aims to shorten hit identification and lead optimization timelines significantly. This milestone is driving the small molecule drug discovery market by accelerating R&D productivity, increasing pharma spending on AI-enabled discovery, and encouraging similar partnerships across the industry.
2. Eli Lilly Expansion of Insilico Medicine Collaboration (2026)
In 2026, Eli Lilly and Company expanded its collaboration with Insilico Medicine in a deal worth up to USD 2.75 billion in milestone payments, strengthening AI-driven small molecule development. The partnership gives Lilly access to AI-designed therapeutic candidates and computational chemistry capabilities to optimize early-stage drug pipelines. This development is driving the market because it demonstrates rising confidence from major pharmaceutical companies in AI-based small molecule discovery, increasing capital inflow into discovery platforms and encouraging biotech-pharma licensing activity to accelerate candidate generation.
3. AstraZeneca–CSPC Strategic Collaboration for AI Small Molecules (2025)
In June 2025, AstraZeneca partnered with CSPC Pharmaceutical Group to develop novel oral small molecules using an AI-driven drug discovery platform. The collaboration focuses on identifying compounds with strong developability and optimized target binding through computational modeling. This milestone is helping drive the market by strengthening cross-border R&D collaborations, expanding AI-assisted molecule screening, and increasing demand for computational chemistry and discovery outsourcing services. It also highlights growing investment in precision therapeutics and oral small-molecule medicines.
4. FDA’s Growing Support for AI in Drug Development (Government Initiative)
The U.S. Food and Drug Administration has increasingly formalized the role of artificial intelligence in drug development, acknowledging a significant rise in drug applications incorporating AI across nonclinical and clinical development stages. The agency’s expanding regulatory framework around AI-supported development is a major milestone because it lowers uncertainty for pharmaceutical firms adopting computational discovery tools. By improving regulatory clarity and encouraging technology integration, the FDA initiative is helping accelerate small molecule discovery workflows, increase confidence in AI-generated candidates, and support faster progression of compounds from discovery to clinical testing.
The small molecule drug discovery market is segmented by region into North America, Europe, Asia-Pacific, Latin America, and LAMEA. Here is a brief overview of each region:
The North America small molecule drug discovery market size was valued at USD 40.01 billion in 2025 and is expected to surpass around USD 92.84 billion by 2035.
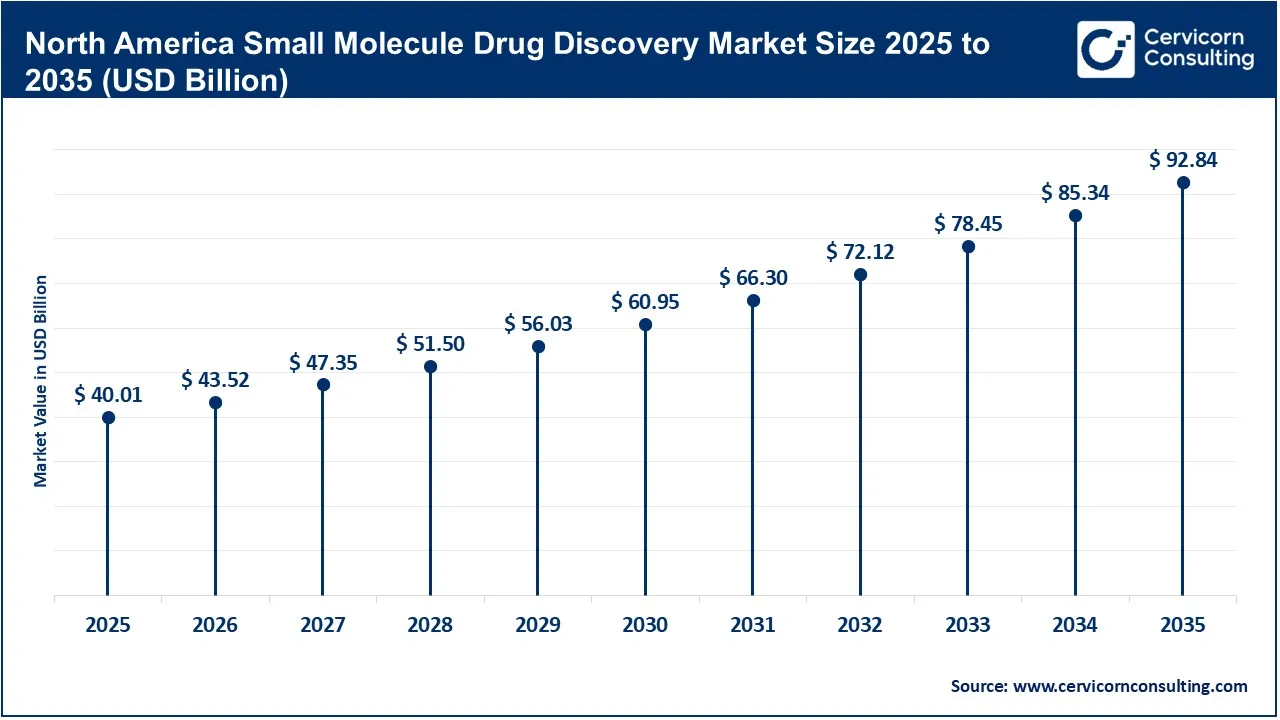
The North America market is highly advanced, supported by strong pharmaceutical research capabilities, extensive biotechnology innovation, and substantial healthcare R&D investments. The region benefits from the presence of major pharmaceutical companies, biotechnology firms, contract research organizations (CROs), and advanced academic research institutes that actively invest in next-generation therapeutics. Rapid adoption of artificial intelligence, computational drug discovery, and high-throughput screening technologies further accelerates early-stage molecule identification and optimization. In addition, favorable regulatory support, strong intellectual property protection, and rising investments in precision medicine continue to strengthen the region’s leadership in small molecule innovation, making North America one of the most mature and innovation-driven markets globally.
United States: Strong pharmaceutical leadership, robust biotechnology ecosystems, high drug R&D spending, and increasing AI-driven drug discovery partnerships drive market expansion.
Canada: Expanding biotechnology research, growing government support for life sciences, and increasing clinical and preclinical drug research activities accelerate market growth.
The Asia-Pacific small molecule drug discovery market size was estimated at USD 20.19 billion in 2025 and is projected to hit around USD 46.85 billion by 2035. The Asia-Pacific market is experiencing strong growth, supported by expanding pharmaceutical manufacturing capabilities, rising biotechnology investments, and increasing outsourcing of drug discovery activities from global pharmaceutical companies. Countries across the region are strengthening their life sciences infrastructure through government-backed healthcare initiatives, growing clinical research ecosystems, and increasing adoption of AI-based drug discovery technologies. The availability of a large patient population, cost-efficient research services, and a rapidly expanding talent pool further position Asia-Pacific as a strategic hub for preclinical research, medicinal chemistry, and contract drug discovery. Increasing investments in oncology, infectious diseases, and precision medicine are also accelerating market development across the region.
China: Strong government support for biotechnology, rapid pharmaceutical innovation, and expanding AI-enabled drug discovery platforms are driving market growth.
India: Cost-efficient R&D services, a strong generic pharmaceutical base, and expanding CRO capabilities support market expansion.
The Europe small molecule drug discovery market size was accounted for USD 26.55 billion in 2025 and is forecasted to grow around USD 61.60 billion by 2035. The Europe market is highly developed, supported by a strong pharmaceutical manufacturing base, advanced biotechnology ecosystems, and extensive public–private research collaborations. The region benefits from the presence of globally recognized pharmaceutical companies, robust academic research institutions, and favorable funding for drug innovation through government and regional initiatives. Increasing investments in precision medicine, oncology therapeutics, and AI-assisted drug discovery are accelerating small molecule development. In addition, strong regulatory frameworks, growing cross-border clinical research, and rising outsourcing partnerships continue to position Europe as a key hub for early-stage drug discovery and medicinal chemistry innovation.
United Kingdom: Robust life sciences ecosystems, strong university research networks, and growing AI-driven drug discovery investments accelerate market expansion.
Germany: Strong pharmaceutical manufacturing capabilities, high R&D spending, and advanced biotechnology research drive market growth.
Small Molecule Drug Discovery Market Share, By Region, 2025 (%)
| Region | Revenue Share, 2025 (%) |
| North America | 42.8% |
| Europe | 28.4% |
| Asia-Pacific | 21.6% |
| LAMEA | 7.2% |
The LAMEA small molecule drug discovery market was valued at USD 6.73 billion in 2025 and is expected to reach around USD 15.62 billion by 2035. The LAMEA market is gradually expanding, supported by improving healthcare infrastructure, rising pharmaceutical investments, and increasing government focus on biotechnology and life sciences development. Countries in the region are strengthening clinical research capabilities and encouraging partnerships with global pharmaceutical companies to enhance local drug development ecosystems. Growing prevalence of chronic diseases, rising demand for affordable therapeutics, and expanding access to healthcare services are creating opportunities for small molecule innovation. Additionally, increasing investments in contract research, generic pharmaceuticals, and technology-enabled discovery platforms are contributing to market development across the region.
Brazil: Expanding pharmaceutical manufacturing, growing biotechnology investments, and increasing clinical research activities support market growth.
Saudi Arabia: Strong healthcare modernization initiatives, rising biotech investments, and government-led diversification programs accelerate market expansion.
The small molecule drug discovery market is segmented into drug type, drug discovery process / workflow, technology, therapeutic area, end user, and geography.
Synthetic small molecules dominate the small molecule drug discovery market due to their well-established manufacturing pathways, cost-effectiveness, scalability, and strong therapeutic versatility across multiple disease areas. These molecules are widely preferred by pharmaceutical companies because they exhibit favorable pharmacokinetic properties, high oral bioavailability, and easier optimization during development. In addition, a majority of approved drugs globally continue to be chemically synthesized, reinforcing their commercial leadership. Their extensive use in oncology, cardiovascular, and infectious disease treatments further supports their dominant market position.
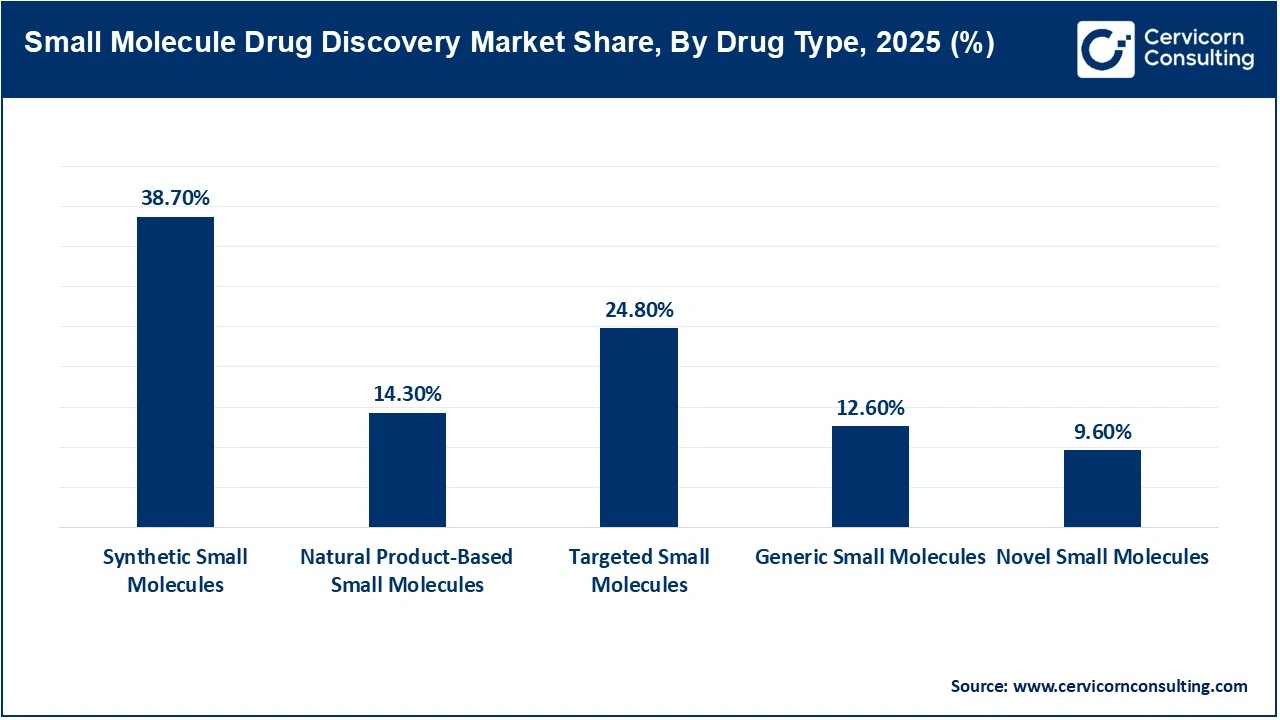
Targeted small molecules are emerging as the fastest-growing segment owing to increasing demand for precision medicine and highly selective therapies with fewer side effects. These molecules are specifically designed to interact with disease-related molecular pathways, making them particularly effective in oncology, autoimmune disorders, and rare diseases. Rising investments in biomarker-driven therapeutics and advances in genomics and molecular profiling are accelerating their adoption. Pharmaceutical companies are increasingly prioritizing targeted therapies to improve treatment efficacy and patient outcomes, contributing to rapid segment expansion.
Lead optimization dominates the market because it represents a critical stage where promising compounds are refined for potency, selectivity, safety, and pharmacokinetic performance before clinical progression. This phase demands substantial investment in medicinal chemistry, toxicity testing, and molecular refinement, making it one of the most resource-intensive areas of drug discovery. Pharmaceutical firms prioritize this process to reduce late-stage failures and improve candidate success rates. Continuous advancements in computational chemistry and predictive modeling are further strengthening the importance of lead optimization in the market.
Global Small Molecule Drug Discovery Market, By Drug Discovery Process / Workflow, 2025 (%)
| Drug Discovery Process / Workflow | Revenue Share, 2025 (%) |
| Target Identification & Validation | 18.4% |
| Hit Generation / Hit Identification | 20.1% |
| Lead Identification | 17.2% |
| Lead Optimization | 27.5% |
| Candidate Validation / Preclinical Development | 16.8% |
Target identification and validation is witnessing the fastest growth due to advances in genomics, proteomics, and AI-powered biological analysis that help uncover novel disease mechanisms. The increasing focus on precision medicine and personalized therapeutics has amplified the need to identify highly specific biological targets before drug development begins. Companies are investing heavily in biomarker research and disease pathway mapping to enhance therapeutic effectiveness and reduce development risks. This early-stage focus is helping improve drug success probabilities and accelerating segment growth.
High-throughput screening remains the dominant technology segment because of its ability to rapidly test thousands to millions of compounds against biological targets in a highly automated manner. Pharmaceutical and biotechnology companies rely heavily on HTS to accelerate hit identification and reduce discovery timelines. Its widespread integration into drug discovery laboratories, combined with advancements in robotics, assay development, and automation, supports sustained demand. HTS continues to play a crucial role in identifying promising molecules across oncology, infectious disease, and CNS drug pipelines.
Global Small Molecule Drug Discovery Market, By Technology, 2025 (%)
| Technology | Revenue Share, 2025 (%) |
| High-Throughput Screening (HTS) | 25.2% |
| Structure-Based Drug Design (SBDD) | 16.4% |
| Fragment-Based Drug Discovery (FBDD) | 10.8% |
| Virtual Screening / In-Silico Drug Discovery | 14.7% |
| Combinatorial Chemistry | 11.5% |
| AI & Machine Learning-Based Drug Discovery | 12.9% |
| Bioinformatics & Computational Modeling | 8.5% |
AI and machine learning-based drug discovery is the fastest-growing technology segment due to its ability to significantly reduce hit-to-lead timelines and improve candidate prediction accuracy. AI platforms can analyze large biological datasets, predict molecular interactions, optimize compounds, and identify potential toxicity risks faster than traditional approaches. Increasing collaborations between pharmaceutical firms and AI startups are accelerating technology adoption. The growing need to reduce R&D costs and shorten drug development timelines is expected to further propel this segment’s expansion.
Oncology dominates the small molecule drug discovery market due to the rising global cancer burden and substantial pharmaceutical investment in targeted cancer therapies. Small molecules are widely used in oncology because of their ability to penetrate cells and inhibit cancer-driving proteins effectively. Increasing approvals of kinase inhibitors and targeted therapies continue to strengthen this segment’s leadership. Additionally, growing research in personalized oncology and biomarker-driven treatment approaches has encouraged sustained innovation and investment, reinforcing oncology’s strong market position.
Global Small Molecule Drug Discovery Market, By Therapeutic Area, 2025 (%)
| Therapeutic Area Segment | Revenue Share, 2025 (%) |
| Oncology | 29.6% |
| Cardiovascular Diseases | 12.8% |
| Neurological Disorders / CNS Diseases | 11.7% |
| Infectious Diseases | 10.9% |
| Respiratory Diseases | 7.1% |
| Metabolic Disorders | 8.6% |
| Immunological / Autoimmune Diseases | 7.9% |
| Gastrointestinal Disorders | 4.5% |
| Rare Diseases | 3.8% |
| Others | 3.1% |
Rare diseases represent the fastest-growing therapeutic segment due to increasing orphan drug incentives, regulatory support, and unmet medical needs. Pharmaceutical companies are increasingly focusing on developing highly targeted small molecules for genetically defined rare conditions, supported by advancements in genomics and precision medicine. Favorable regulatory policies, including expedited approvals and exclusivity benefits, are encouraging greater investment in orphan therapeutics. Rising patient advocacy and improved diagnostic capabilities are also contributing to accelerated growth within this segment.
Pharmaceutical companies dominate the market because they account for the majority of global spending on drug discovery, clinical research, and commercialization activities. Large pharmaceutical firms possess substantial R&D budgets, advanced laboratory infrastructure, and extensive compound libraries that enable continuous small molecule innovation. Their strong focus on oncology, metabolic disorders, and neurological diseases sustains demand for advanced discovery technologies. Strategic collaborations with AI companies, biotech firms, and CROs also strengthen their leading role in market expansion.
Small Molecule Drug Discovery Market, By End User, 2025 (%)
| End User | Revenue Share, 2025 (%) |
| Pharmaceutical Companies | 46.8% |
| Biotechnology Companies | 21.9% |
| Contract Research Organizations (CROs) | 18.6% |
| Academic & Research Institutes | 8.2% |
| Government Research Organizations | 4.5% |
Contract research organizations are the fastest-growing end-user segment due to rising outsourcing trends among pharmaceutical and biotechnology companies seeking cost efficiency and faster development timelines. CROs provide specialized expertise in medicinal chemistry, screening, lead optimization, and preclinical services, enabling companies to streamline operations and reduce infrastructure costs. Increasing complexity in drug discovery and pressure to improve productivity are driving greater reliance on outsourced discovery models. Expanding global R&D partnerships are further accelerating growth in this segment.
By Drug Type
By Drug Discovery Process / Workflow
By Technology
By Therapeutic Area
By End User
By Geography