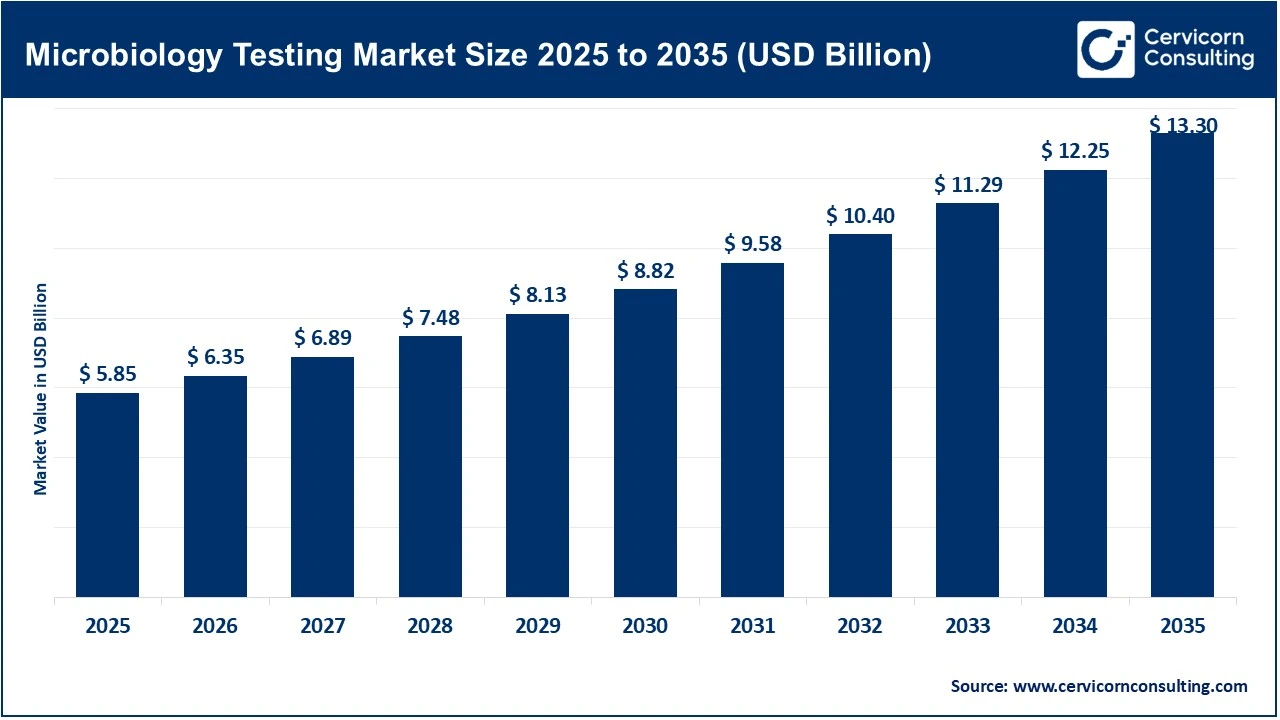
The global microbiology testing market size reached at USD 5.85 billion in 2025 and is expected to be worth around USD 13.30 billion by 2035, exhibiting a compound annual growth rate (CAGR) of 8.6% over the forecast period from 2026 to 2035. The microbiology testing market growth is the high burden of infectious diseases worldwide, which demands frequent and specialized testing. Diseases such as tuberculosis, malaria, and various sexually transmitted infections remain major public health issues, while new zoonotic threats and seasonal viral outbreaks perpetually drive demand. According to the World Health Organization, infectious diseases cause a significant share of global deaths, with over 10 million fatalities annually. The organization also reported that tuberculosis alone resulted in about 1.3 million deaths globally in recent years, emphasising the ongoing need for effective diagnostic systems. Global health organizations state that infectious diseases continue to account for a substantial percentage of worldwide mortality, especially in low- and middle-income countries, projecting a market value of USD 7 billion by the end of the decade.
The rapid adoption of advanced diagnostic technologies in clinical settings is another key growth factor, as it greatly enhances the speed and accuracy of pathogen detection. The shift from slow, culture-based methods to rapid molecular assays has revolutionised patient management, particularly in cases of septicemia and respiratory distress, where quick intervention is critical. Molecular diagnostic tests can cut pathogen detection time by up to 70–80% compared to traditional methods, significantly improving treatment outcomes. Moreover, diagnostic platforms utilizing nanotechnology that offer sub-picomolar sensitivity enable early detection of pathogens at the initial stages of infection.
Automation and Robotic Technology in Microbiology Testing Market
One of the biggest trends in the microbiology testing market is the rapid shift toward Total Laboratory Automation (TLA) and collaborative robotics. Clinical laboratories are increasingly using hands-free automated systems to handle rising testing volumes while reducing human intervention. Leading companies like Roche and BD are launching automotive systems that lower human error and provide faster turnaround times, especially as labor shortages worsen. Additionally, clinical labs are seeing notable drops in error rates and labor needs, while stabilising workflow processes even during busy periods. Automation boosts operational efficiency by up to 30%, thereby shortening result reporting times and improving patient triage.
What is Microbiology Testing?
Microbiology testing involves examining samples such as blood, water, or food to detect tiny organisms, including bacteria, viruses, and fungi. It helps doctors and scientists identify the causes of infections or contamination. These tests are widely used in hospitals, laboratories, and industries to ensure safety and effective treatment. By detecting harmful microbes early, microbiology testing plays a crucial role in safeguarding health and preventing disease spread.
What are the key Applications sectors of Microbiology Testing?
1. Clinical Microbiology and Healthcare Diagnostics
In healthcare, microbiology testing is essential for diagnosing infections ranging from sepsis to respiratory illnesses. Standardized guidelines in clinical microbiology ensure that specimen collection and subsequent transport across settings are performed properly to maintain sample quality. Preserving sample integrity to obtain accurate results is crucial in clinical microbiology. Rapid diagnostics are also vital for antimicrobial stewardship, helping to narrow therapy and enhance patient outcomes.
2. Industrial Food Safety and Quality Assurance
In the food sector, microbiology focuses on detecting foodborne pathogens such as Shiga toxin-producing Escherichia coli (STEC) to prevent illnesses caused by contaminated food. All STEC strains are considered potentially pathogenic, necessitating testing for STEC in meat, dairy, and produce, among other foods. Regulatory frameworks, such as ISO standards, are established to standardize rapid detection methods for ensuring food safety.
3. Environmental Monitoring and Water Quality Testing
In the environmental sector, microbiology testing focuses on monitoring indicators of fecal contamination, such as E. coli, to ensure the safety of drinking water. For example, molecular methods are increasingly used alongside culture-based techniques to provide a more comprehensive assessment of microbial risks, especially those caused by enteric viruses and protozoa.
Report Scope
| Area of Focus | Details |
| Market Size in 2026 | USD 6.35 Billion |
| Market Size in 2035 | USD 13.30 Billion |
| CAGR from 2026 to 2035 | 8.60% |
| Dominant Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Key Segments | Product, Test Type, Application, End User, Region |
| Key Companies | Hologic Inc., Shimadzu Corporation, Thermo Fisher Scientific, bioMérieux, Merck KGaA, Becton, Dickinson and Company, Neogen Corporation, Abbott Laboratories, Danaher Corporation, Bio-Rad Laboratories, QIAGEN, Agilent Technologies, Bruker Corporation, F. Hoffmann-La Roche |
1. Corporate Expansion and Strategic Diagnostic Acquisitions
Corporate expansion and strategic acquisitions are crucial in the growth of the microbiology testing market. Leading diagnostic companies are increasingly investing in multi-billion dollar acquisitions to strengthen "sample-to-result" workflows and develop integrated diagnostic ecosystems. The acquisition of specialized molecular diagnostic firms like Danaher and Thermo Fisher supports rapid testing strategies for their current laboratory setups, leveraging rapid diagnostic capabilities. These acquisitions usually range from USD 1 billion to USD 3 billion, as proprietary molecular technologies command a premium. Consequently, market consolidation has increased the prevalence of integrated diagnostic platforms that encompass extraction, amplification, and detection, contributing to a 15% growth in automated systems.
2. Government Funding and Public Health Surveillance Programs
Government funding and health surveillance programs are major drivers of the market. Growing global concern over antimicrobial resistance (AMR) has led to significant investments in diagnostic activities. For example, in 2025, global health agencies announced funding of over USD 500 million for diagnostic stewardship to develop tests that accurately identify viral or bacterial infections. This funding aims to reduce the over-prescription of antibiotics, which contributes to AMR. Additionally, the United States Department of Agriculture introduced regulatory frameworks, such as the New Swine Inspection Standards (NSIS), that require improved pathogen testing, including quantitative Salmonella analysis of pork products. These initiatives support broader public health goals, such as the Healthy People 2030 program, further boosting demand for microbiology testing solutions.
3. Breakthroughs in Regulatory Approvals for Novel Assays
Recent advancements in the regulatory framework have led to more approvals for "syndromic testing" panels and changed how infectious diseases are diagnosed. In late 2024 and early 2025, the U.S. Food and Drug Administration approved next-generation multiplex assays capable of detecting more than 20 respiratory or gastrointestinal pathogens in a single test. Clinical studies show that these innovations can reduce the time to sepsis diagnosis from an average of 48 hours to less than 6 hours. After these approvals, the use of multiplex panels in level 1 trauma centers increased by over 20% shortly afterwards.
4 Global Diagnostic Infrastructures via Strategic Partnerships
Strategic partnerships are helping expand the global diagnostics infrastructure, marking a significant milestone for the markets. Collaboration efforts focus on deploying portable, solar-powered molecular diagnostic units in regions such as sub-Saharan Africa and Southeast Asia to combat endemic diseases such as tuberculosis and malaria. Often funded by international grants, these organisations have the potential to establish a sustainable diagnostic infrastructure capable of responding to regional outbreaks. Likewise, partnerships between AI developers and diagnostic manufacturers aim to accelerate the adoption of cloud-based microbiology data platforms, enabling real-time sharing of pathogen genomic information across different regions.
The microbiology testing market is segmented by region into North America, Europe, Asia-Pacific, and LAMEA. Here is a brief overview of each region:
The Asia-Pacific microbiology testing market size was accounted for USD 1.64 billion in 2025 and is forecasted to grow around USD 3.72 billion by 2035. The Asia Pacific region is the fastest-growing market, driven by significant investments in healthcare infrastructure and a high burden of infectious diseases. Countries such as China and India are emerging as global leaders in biotechnology research and financing, especially at the intersection of AI and microbial applications. The main trend in the region is the decentralization of diagnostic services, with testing capabilities increasingly being brought closer to patients, often at Tier 2 and Tier 3 levels of care. Additionally, rising rates of respiratory infections and gastrointestinal disorders will further drive the demand for routine microbiology screening.
China is leading regional growth through Massive Decentralization of Healthcare
China is rapidly growing its diagnostic system by enhancing hospital laboratories in smaller cities to ease the load on major cities.
India to Emerge as a Global Centre for Pharmaceutical Quality Control Testing
India is significantly impacted by the need for strict quality control in pharmaceutical manufacturing.
The North America microbiology testing market size was estimated at USD 2.52 billion in 2025 and is predicted to surpass around USD 5.72 billion by 2035.

North America holds the largest share of the microbiology testing market, supported by a highly advanced healthcare ecosystem and a strong focus on research and development. The main advantages in this region include the early adoption of rapid molecular diagnostics and automated laboratory tracking systems, which are essential components of the precision medicine model. High healthcare spending and the presence of many industry leaders facilitate a swift shift from traditional diagnostic methods to syndromic panels. Additionally, in agricultural regions, the integration of sensor technologies and microbial bio-inoculants serves a clinical purpose, leading to crop yields increasing by over 25% under optimized conditions.
The United States is a dominant country in the market with High Investment in R&D and a focus on Precision Medicine
The U.S. market features a high concentration of diagnostic innovators and a healthcare system that promotes rapid and precise testing.
Canada is continuous focus on biopharmaceutical research and protecting environmental resources.
Canada's microbiology market highlights its strong emphasis on biopharmaceutical research and development (R&D) and on preserving its valuable natural resources.
The Europe microbiology testing market size was valued at USD 1.40 billion in 2025 and is expected to reach around USD 3.19 billion by 2035. Europe market is shaped by a strong regulatory environment focused on ensuring patient safety and protecting the environment. The introduction of the In Vitro Diagnostic Regulation (IVDR) has raised the standard for clinical evidence and accuracy in diagnostics, forcing manufacturers to modernize their portfolios. Notably, Europe also remains a leader in food safety microbiology, as it maintains strict EU regulations and legislation for monitoring foodborne pathogens. The region is increasingly seeing specialized testing of alternative sources of protein, as the insect farming industry is encountering several challenges with unnoticed microbial infections that can lead to extreme economic loss if not detected early.
The United Kingdom is a leader in Genomic Surveillance and Integration with Public Health.
The U.K.'s focus on antimicrobial resistance (AMR) is increasing engagement with rapid susceptibility testing technologies.
Germany drives industrial automation and clinical efficiency
Germany is a major industrial centre in Europe, merging advanced precision engineering with large-scale clinical diagnostic activities.
Microbiology Testing Market Share, By Region, 2025 (%)
| Region | Revenue Share, 2025 (%) |
| North America | 43% |
| Europe | 24% |
| Asia Pacific | 28% |
| LAMEA | 9% |
The LAMEA microbiology testing market was valued at USD 0.53 billion in 2025 and is anticipated to hit around USD 1.20 billion by 2035. The LAMEA market is driven by the modernization of laboratory facilities and efforts to increase access to healthcare in emerging economies. The Middle East, including countries such as the UAE and Saudi Arabia, is heavily investing in diagnostic technology, with the construction of high-tech diagnostic hubs to boost their growing medical tourism industries and provide world-class clinical services. In Latin America, the focus is on strengthening public health surveillance systems and controlling endemic infectious diseases like Zika and Dengue.
Brazil is a country that utilizes Agricultural Microbiology and Infectious Disease Management
Brazil navigates the balance between a need for advanced clinical diagnostics and a substantial market in the global-leading agricultural microbiology sector.
United Arab Emirates to Enhance Medical Tourism via High-Tech Diagnostics
The UAE is establishing itself as a top medical tourism destination by investing in advanced diagnostic infrastructure.
The microbiology testing market is segmented into product, test type, application, end user, and region.
Reagents constitute the largest product segment in the microbiology testing market, mainly due to the high frequency of routine clinical testing and the heavy reliance on specialized consumables for every diagnostic assay. In the diagnostic sector, reagents generate a steady revenue stream that is essential for supporting manufacturers. Laboratories must continuously purchase kits for culture media, stains, and molecular probes to sustain ongoing testing procedures.
Microbiology Testing Market Share, By Product, 2025 (%)
| Product | Revenue Share, 2025 (%) |
| Reagents | 75% |
| Instruments | 25% |
Instruments are the fastest-growing segment in the market, primarily driven by the increasing shift toward laboratory automation and the demand for high-throughput capabilities. Laboratories face immense pressure to deliver faster results with fewer staff, leading to the rapid adoption of advanced technologies such as automated plate-streakers, high-speed incubators, and fully integrated “sample-to-result” molecular platforms. Moreover, the integration of artificial intelligence (AI) and robotics into laboratory systems is improving efficiency and propelling the growth of high-value instrument installations.
Bacterial testing holds the largest market share, mainly due to the high prevalence of common bacterial infections, such as urinary tract infections, streptococcal pharyngitis, and foodborne illnesses caused by pathogens like C. botulinum and Salmonella. Traditional bacterial culture methods continue to form the basis of microbiology testing in many clinical laboratories, where the high volume of these routine tests considerably contributes to daily laboratory workloads worldwide.
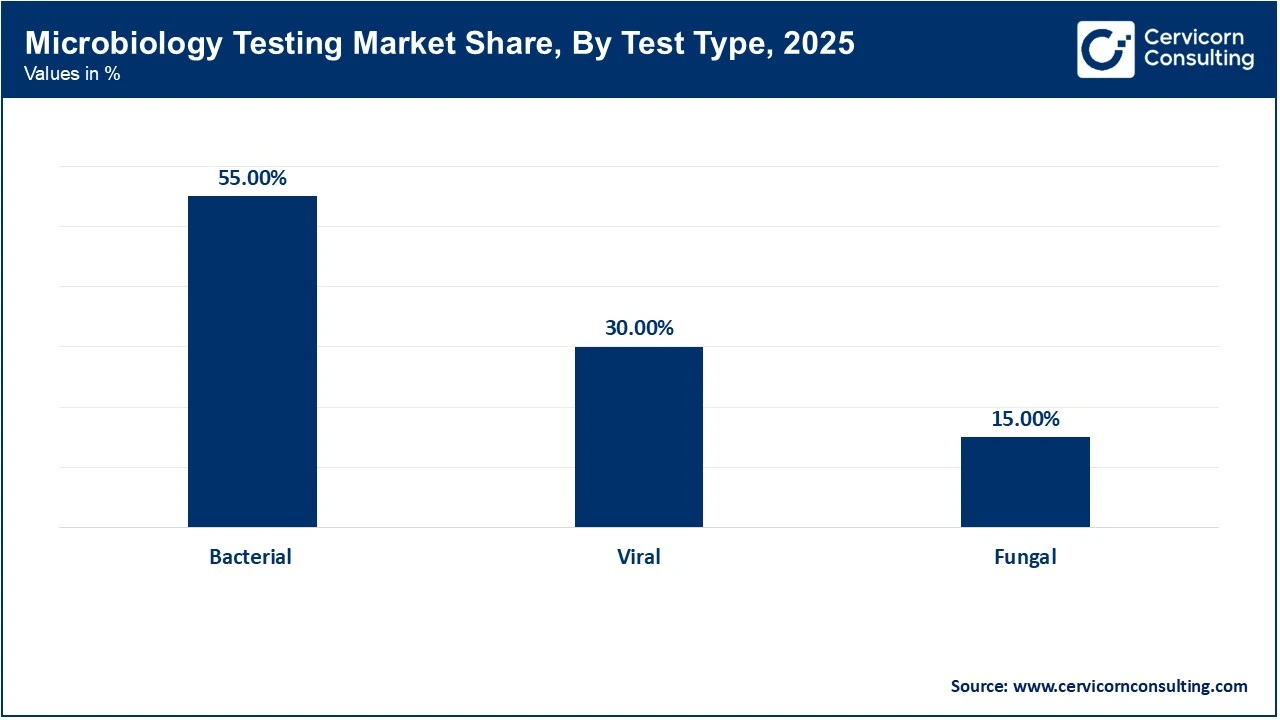
Viral testing is the fastest-growing segment in the market due to advances in molecular diagnostics and the increasing global focus on viral disease detection. There has been a permanent shift in public health infrastructure towards viral screening since the COVID-19 pandemic. Technologies such as PCR and NGS are now widely used to detect respiratory viruses such as influenza and RSV, as well as blood-borne pathogens. Viral tests have expanded the market through rapid, point-of-care tests in non-traditional settings.
Gastrointestinal diseases account for the largest share of the microbiology testing market due to the high global incidence of enteric infections and strict food safety regulations. Regular screening for pathogens such as E. coli and Salmonella is crucial in both clinical and industrial settings. Gastrointestinal testing usually involves multiple stool cultures and molecular tests performed daily to control outbreaks and support public health. The ongoing need to screen for enteric pathogens in food and beverage facilities further confirms gastrointestinal applications as the top revenue-driving segment in the market.
Microbiology Testing Market Share, By Application, 2025 (%)
| Application | Revenue Share, 2025 (%) |
| Gastrointestinal Diseases | 30% |
| Respiratory Diseases | 20% |
| Bloodstream Infection | 16% |
| Sexually Transmitted Diseases | 12% |
| Urinary Tract Infections | 10% |
| Periodontal Diseases | 5% |
| Others | 7% |
Respiratory diseases are the fastest-growing area in the market due to rising awareness of respiratory health and the use of syndromic testing for bacterial and viral pneumonia. In the post-pandemic era, respiratory diagnostics have increasingly relied on multiplex panels that can detect multiple pathogens from a single swab, simplifying the diagnostic process for symptomatic patients. The shift toward rapid molecular respiratory testing is vital for reducing the spread of infectious diseases and better managing hospital isolation resources.
Hospitals lead in the microbiology testing market because they are the primary care points for patients with acute infections. These environments require well-equipped in-house microbiology laboratories to enable prompt diagnosis and effective treatment, as well as to control healthcare-associated infections (HAIs). Large hospital networks are also major investors in lab automation, aiming to improve efficiency, scalability, and testing capacity.
Microbiology Testing Market Share, By End User, 2025 (%)
| End User | Revenue Share, 2025 (%) |
| Hospitals | 48% |
| Diagnostic Centers | 28% |
| Academic and Research Institutes | 16% |
| Others | 8% |
Academic and research institutions are the fastest-growing end-user segments, driven by increased global funding for infectious disease research and pandemic preparedness. These institutions are early adopters of advanced technologies like Next-Generation Sequencing (NGS) and CRISPR-based diagnostics, which emerged before widespread clinical use.
By Product
By Test Type
By Application
By End User
By Region