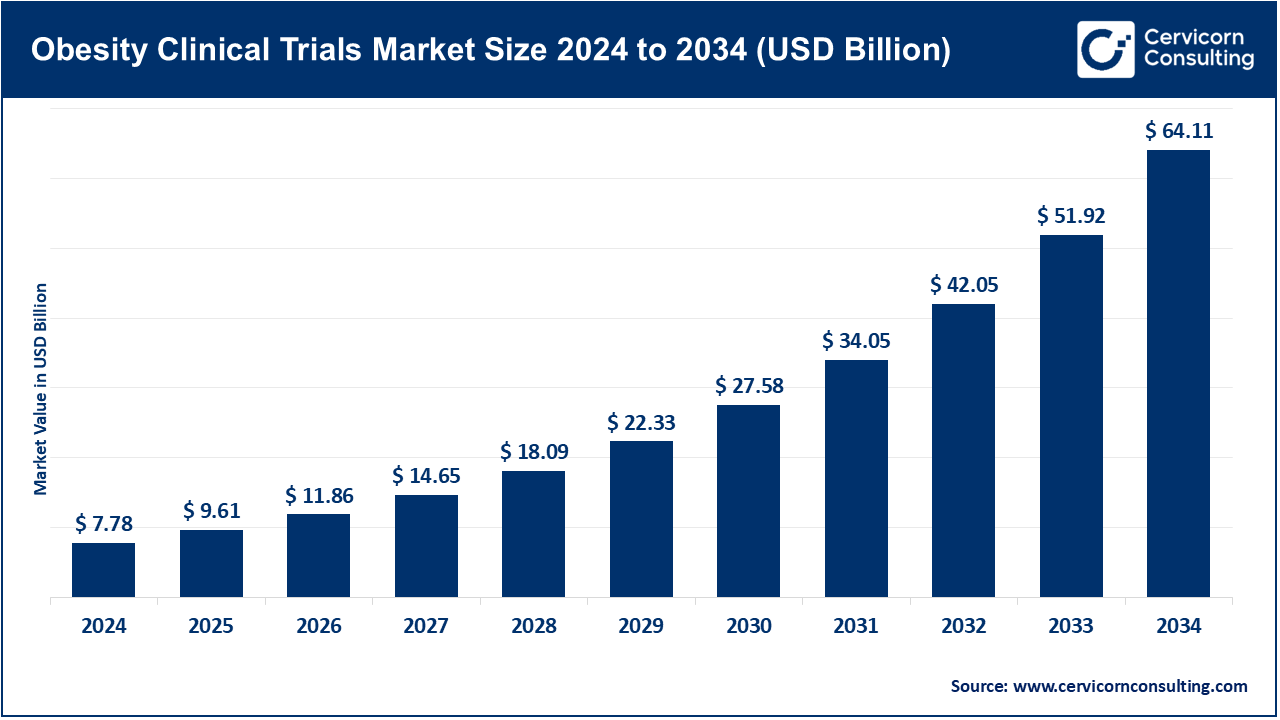
The global obesity clinical trials market size was valued at USD 7.78 billion in 2024 and is anticipated to reach around USD 64.11 billion by 2034, growing at a compound annual growth rate (CAGR) of 23.48% over the forecast period from 2025 to 2034.
The rise in obesity around the world has led to a major public health crisis. Sedentary lifestyles, ultra-processed diets, and more people living in cities are largely to blame. This growing problem is a key driver of activity in clinical trials. The World Health Organization reported that in 2022, around 2.5 billion adults were marked as overweight, and nearly 890 million of those reached the level of obesity. If trends continue, this figure could soar to 4 billion by 2035. Obesity frequently coexists with serious health conditions such as type 2 diabetes, cardiovascular disease, and various mental health disorders. In response, pharmaceutical developers are increasingly focusing on multi-indication trials that assess new therapies not only for weight loss but also for managing these comorbidities. An ICON survey showed more than 64% of participants think mixing treatment methods will shape future obesity care. When asked what the main focus should be, 55% chose diabetes, 48% highlighted metabolic disorders, and 38% pointed to heart and blood vessel conditions. These interconnected health needs enhance the opportunity for clinical trials and expedite the development of new treatments.
What is Obesity Clinical Trials?
Obesity affects approximately 1 billion people and is a priority in clinical research. This problem is closely associated with health problems such as type 2 additional metabolism problem fatty liver disease and heart disease. Recent progress in medicines, such as GLP-1 receptor agonists semaglutide (Wegovy) and tirzepatide (Zepbound), have transformed the way of treating obesity. Not only have these new therapies improved the outlook for patients, but they have also given rise to a surge in the number of clinical trials. Both large pharmaceutical companies and biotech startups are entering the field. They are driving the development of next-generation therapies. This includes dual and triple agonists, oral formulations, and combination treatments. All these aims to offer better solutions for those dealing with obesity.
Obesity Clinical Trials
| Year | New Obesity Trials Registered | Estimated Total Active Trials |
| 2020 | 180 | 600 |
| 2021 | 210 | 700 |
| 2022 | 250 | 850 |
| 2023 | 300 | 1000 |
| 2024 | 320 | 1100 |
Phase I: The major objective of Phase I studies is to assess safety, tolerability, and pharmacokinetics of new anti-obesity agents. Studies in small healthy volunteer populations or obese volunteer populations make an integral part of early validation but constitute a relatively smaller portion of the market because there are very few such studies and they run for a shorter duration compared to later phase studies.
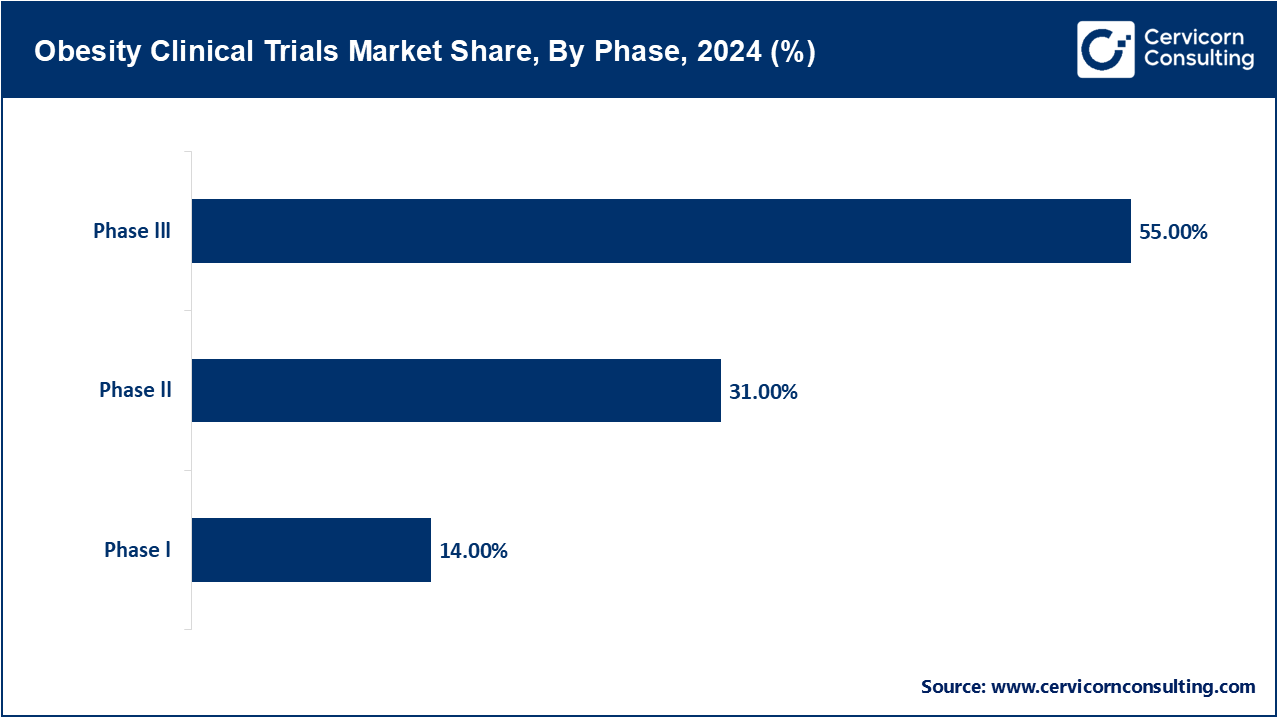
Phase II: Presently, there has been a great uptick of phase II trials since in this phase, the efficacy of the drug is being assessed together with dosage optimization on a larger group of the population with obesity. In turn, this helps determine which dosages of the investigational drugs may be most effective and safe. Currently, there is great development regarding GLP-1 as well as GIP agonists besides new oral formulations and combination therapies. The successful medications semaglutide and tirzepatide have thus opened up an avenue/pipeline/floodgate for increased amounts of phase II studies to test next generation dual and triple agonists promised to bring efficacy with lesser side effects.
Phase III: In Phase III clinical trials, new therapies for obesity are tested for safety and effectiveness in large, multicultural populations, with 3000 to 5000 participants for the 12–18-month study period. These trials are essential to validating the effectiveness of new therapies, to tracking unwanted side effects, and to comparing new treatments with the current standard of care. Leading pharmaceutical companies, including Novo Nordisk, Eli Lilly, and Amgen, are currently engaged in multiple ongoing Phase III studies globally to support submissions to regulatory authorities and pursue market access.
Monotherapy for Obesity: Monotherapy for obesity is the use of one pharmaceutical agent in assisting patients diagnosed with obesity to reduce and maintain weight loss. This has traditionally been a less active area of clinical investigations until recently over the past decade primarily due to newer agents such as GLP-1 receptor agonists. These medications, semaglutide (Wegovy) and liraglutide (Saxenda) have indicated effectiveness as monotherapies resulting in average weight reduction of about 10-15% in clinical studies which shifted the perception that obesity treatment should only involve pharmacologic management after not achieving adequate results from lifestyle interventions. The positive outcome of these medications catalyzed further research into akin and next-gen therapies including tirzepatide (Zepbound) as well as trial drugs like AMG 133, pemvidutide, and CT-388.
Obesity Clinical Trials Market Share, By Indication, 2024 (%)
| Indication | Revenue Share, 2024 (%) |
| Monotherapy for obesity | 40% |
| Obesity with comorbid conditions | 60% |
Obesity with Comorbid Conditions: Many clinical trials today focus on obesity and type 2 diabetes. These conditions often occur together and share metabolic pathways. Researchers are studying medications like tirzepatide and semaglutide for their potential to help with weight loss and improve blood sugar control. This has attracted significant interest from pharmaceutical companies and regulatory agencies. Semaglutide (Wegovy/ Ozempic) and tirzepatide (Mounjaro) are leading the GLP-1 receptor agonists thus capturing 60 to 70 percent of the market share in obesity pharmacotherapy because of their efficacy in weight management and glycemic control. Semaglutide was the largest selling obesity drug in 2024, capturing nearly 40% of sales and was quickly adopted in the US and Europe.
Interventional Studies: Interventional studies are the studies that actually test new anti-obesity therapies by actively assigning interventions to participants. Most of the clinical trials for obesity management are centered on new and innovative pharmacotherapy, like GLP-1 receptor agonist (semaglutide) and dual agonist (tirzepatide) as well as the new combination therapies, and are randomized, double-blind, placebo-controlled trials (RCTs). These trials are designed to assess weight maintenance (often >15% body weight reduction), metabolic changes (HbA1c, lipid profiles), and long-term cardiovascular risks. Important examples are the SURMOUNT and STEP trial programs, landmark RCTs with proven significant benefits that have provided a strong evidence base for regulatory and clinical adoption.
Obesity Clinical Trials Market Share, By Study Design, 2024 (%)
| Study Design | Revenue Share, 2024 (%) |
| Interventional studies | 70% |
| Observational studies | 22% |
| Expanded access studies | 8% |
Observational studies: In such studies, participants are observed in their natural settings and does not involve any active intervention. Such studies have gained immense importance since the burning of understanding the natural course of obesity, patient behavior and adherence patterns, and long-term effects of lifestyle changes or approved therapies.
Expanded Access Studies: One smaller part of the whole are expanded access studies, which allow patients with severe or stubborn obesity to try out new treatments that are not part of formal clinical trials. This is particularly true when there’s no good choice. These trials offer a way for people to access the drug in a compassionate-use setting and provide valuable safety information before it gains full regulatory approval.
The Asia-Pacific region is becoming the fastest-growing place. Obesity rates have gone up a lot, especially in countries like China, India, Japan, and South Korea. China now has more than 180 million adults with obesity, India more than 135 million, and both Japan and South Korea have recorded at least a 30% rise in obesity over the past 20 years. This is because cities are growing quickly, diets are changing, and people are becoming more sedentary. These countries have a lot of patients who aren't being used to their full potential, which makes them appealing to global sponsors who want to add more trial sites. The area also has low-cost operational environments and is seeing improvements in clinical infrastructure. These countries' governments are starting to put more money into research on obesity and public health programs, which will help the economy grow even more.
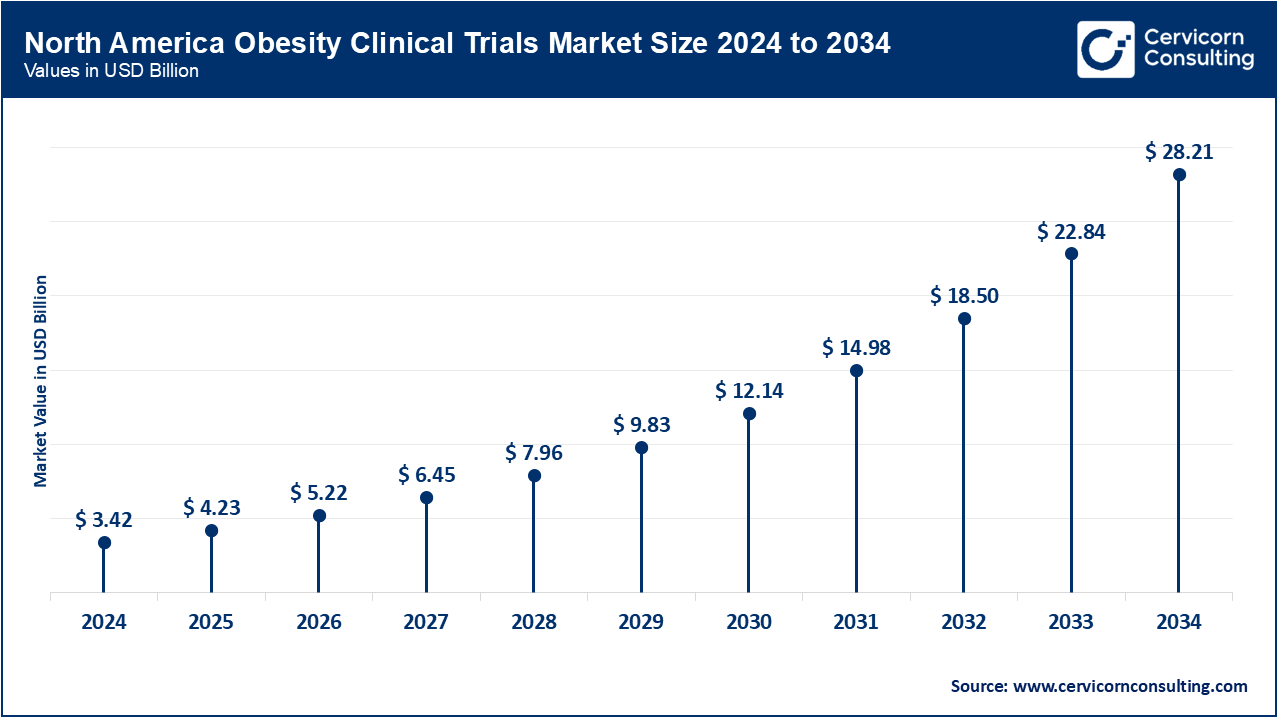
The United States remains the main hub for clinical trials in obesity medication in North America. The strength of our status rests heavily on two strong factors: more than 42 of every 100 adults in the country are now considered obese, and we have an experienced clinical research system already in place. America has state-of-the-art medical facilities, well known CRO’s and a good regulatory environment from the FDA. There is also substantial investment in R&D, for example, Eli Lilly, Pfizer, Novo Nordisk pumping billions into research creates a strong pipeline of trials and planned trials. These drugs have set new benchmarks in obesity treatment, achieving notable weight loss while ensuring solid safety profiles. Meanwhile, key players like Pfizer, AstraZeneca, and Amgen are actively developing their own obesity treatment pipelines. Many of these companies are exploring new oral formulations and are interested in creating dual or triple-action medications. For example, Pfizer is investigating next-generation oral GLP-1 candidates, while Amgen is making progress with a new GLP-1/GIP receptor agonist that has shown encouraging results in early trials. Furthermore, a wave of biotech startups, such as Verve Therapeutics, Metsera, and Structure Therapeutics, is entering the market. These companies are gaining significant venture capital investment and are focusing on innovative methods that target genetic pathways and metabolic signaling to address obesity more effectively.
Obesity Clinical Trials Market Share, By Region, 2024 (%)
| Region | Revenue Share, 2024 (%) |
| North America | 44% |
| Europe | 26% |
| Asia-Pacific | 20% |
| LAMEA | 10% |
Europe ranks second in the world for obesity clinical research, making up about 25 to 28% of all trials happening worldwide. This creates a supportive environment for obesity research. The region focuses on preventive healthcare and lifestyle intervention studies, which matches the goals of obesity trials. Additionally, EU-funded projects and public-private partnerships are improving clinical capabilities across the continent. The field of obesity clinical trials is changing quickly. This shift is mainly due to more research and development, new treatments, and strong competition between large pharmaceutical companies and agile biotech startups. Currently, Novo Nordisk and Eli Lilly are at the forefront with their GLP-1 receptor agonists: semaglutide, known as Wegovy, and tirzepatide, marketed as Mounjaro/Zepbound.
Latin America is now increasingly being considered as a possible market for trials focusing on obesity, particularly in Brazil and Mexico. The increasing level of obesity in such countries is forcing governmental regulations and increasing the demand for therapies that work. Clinical trial expenditure in Latin America is also lower and patient recruitment generally more rapid because there is a high need for treatments and limited access to advanced therapies. Middle East and Africa are the regions, where countries such as, Saudi Arabia, UAE and South Africa obesity figures are on the rise, due to lifestyle changes and economic growth. In the Middle East and Africa, places such as Saudi Arabia, the UAE, and South Africa are watching obesity numbers rise. In Saudi Arabia, one in three adults is now classified as obese, and in the UAE the rate has passed 27%.
Market Segmentation
By Phase
By Indication
By Study Design
By Region
Chapter 1. Market Introduction and Overview
1.1 Market Definition and Scope
1.1.1 Overview of Obesity Clinical Trials
1.1.2 Scope of the Study
1.1.3 Research Timeframe
1.2 Research Methodology and Approach
1.2.1 Methodology Overview
1.2.2 Data Sources and Validation
1.2.3 Key Assumptions and Limitations
Chapter 2. Executive Summary
2.1 Market Highlights and Snapshot
2.2 Key Insights by Segments
2.2.1 By Phase Overview
2.2.2 By Indication Overview
2.2.3 By Study Design Overview
2.3 Competitive Overview
Chapter 3. Global Impact Analysis
3.1 Russia-Ukraine Conflict: Global Market Implications
3.2 Regulatory and Policy Changes Impacting Global Markets
Chapter 4. Market Dynamics and Trends
4.1 Market Dynamics
4.1.1 Market Drivers
4.1.1.1 Increased funding and strategic collaboration
4.1.1.2 Growing demand for non-invasive therapeutic alternatives
4.1.2 Market Restraints
4.1.2.1 High cost and complexity of clinical trials
4.1.2.2 Patient recruitment and retention
4.1.3 Market Challenges
4.1.3.1 Biological Complexity of Obesity as a Disease
4.1.3.2 Regulatory Ambiguity and Shifting Approval Standards
4.1.4 Market Opportunities
4.1.4.1 Growing Focus on Preventive Healthcare
4.1.4.2 Expanding Clinical Research into Emerging Markets
4.2 Market Trends
Chapter 5. Premium Insights and Analysis
5.1 Global Obesity Clinical Trials Market Dynamics, Impact Analysis
5.2 Porter’s Five Forces Analysis
5.2.1 Bargaining Power of Suppliers
5.2.2 Bargaining Power of Buyers
5.2.3 Threat of Substitute Products
5.2.4 Rivalry among Existing Firms
5.2.5 Threat of New Entrants
5.3 PESTEL Analysis
5.4 Value Chain Analysis
5.5 Product Pricing Analysis
5.6 Vendor Landscape
5.6.1 List of Buyers
5.6.2 List of Suppliers
Chapter 6. Obesity Clinical Trials Market, By Phase
6.1 Global Obesity Clinical Trials Market Snapshot, By Phase
6.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2034
6.1.1.1 Phase I
6.1.1.2 Phase II
6.1.1.3 Phase III
Chapter 7. Obesity Clinical Trials Market, By Indication
7.1 Global Obesity Clinical Trials Market Snapshot, By Indication
7.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2034
7.1.1.1 Monotherapy for obesity
7.1.1.2 Obesity with comorbid conditions
Chapter 8. Obesity Clinical Trials Market, By Study Design
8.1 Global Obesity Clinical Trials Market Snapshot, By Study Design
8.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2034
8.1.1.1 Interventional studies
8.1.1.2 Observational studies
8.1.1.3 Expanded access studies
Chapter 9. Obesity Clinical Trials Market, By Region
9.1 Overview
9.2 Obesity Clinical Trials Market Revenue Share, By Region 2024 (%)
9.3 Global Obesity Clinical Trials Market, By Region
9.3.1 Market Size and Forecast
9.4 North America
9.4.1 North America Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.4.2 Market Size and Forecast
9.4.3 North America Obesity Clinical Trials Market, By Country
9.4.4 U.S.
9.4.4.1 U.S. Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.4.4.2 Market Size and Forecast
9.4.4.3 U.S. Market Segmental Analysis
9.4.5 Canada
9.4.5.1 Canada Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.4.5.2 Market Size and Forecast
9.4.5.3 Canada Market Segmental Analysis
9.4.6 Mexico
9.4.6.1 Mexico Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.4.6.2 Market Size and Forecast
9.4.6.3 Mexico Market Segmental Analysis
9.5 Europe
9.5.1 Europe Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.5.2 Market Size and Forecast
9.5.3 Europe Obesity Clinical Trials Market, By Country
9.5.4 UK
9.5.4.1 UK Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.5.4.2 Market Size and Forecast
9.5.4.3 UKMarket Segmental Analysis
9.5.5 France
9.5.5.1 France Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.5.5.2 Market Size and Forecast
9.5.5.3 FranceMarket Segmental Analysis
9.5.6 Germany
9.5.6.1 Germany Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.5.6.2 Market Size and Forecast
9.5.6.3 GermanyMarket Segmental Analysis
9.5.7 Rest of Europe
9.5.7.1 Rest of Europe Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.5.7.2 Market Size and Forecast
9.5.7.3 Rest of EuropeMarket Segmental Analysis
9.6 Asia Pacific
9.6.1 Asia Pacific Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.6.2 Market Size and Forecast
9.6.3 Asia Pacific Obesity Clinical Trials Market, By Country
9.6.4 China
9.6.4.1 China Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.6.4.2 Market Size and Forecast
9.6.4.3 ChinaMarket Segmental Analysis
9.6.5 Japan
9.6.5.1 Japan Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.6.5.2 Market Size and Forecast
9.6.5.3 JapanMarket Segmental Analysis
9.6.6 India
9.6.6.1 India Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.6.6.2 Market Size and Forecast
9.6.6.3 IndiaMarket Segmental Analysis
9.6.7 Australia
9.6.7.1 Australia Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.6.7.2 Market Size and Forecast
9.6.7.3 AustraliaMarket Segmental Analysis
9.6.8 Rest of Asia Pacific
9.6.8.1 Rest of Asia Pacific Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.6.8.2 Market Size and Forecast
9.6.8.3 Rest of Asia PacificMarket Segmental Analysis
9.7 LAMEA
9.7.1 LAMEA Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.7.2 Market Size and Forecast
9.7.3 LAMEA Obesity Clinical Trials Market, By Country
9.7.4 GCC
9.7.4.1 GCC Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.7.4.2 Market Size and Forecast
9.7.4.3 GCCMarket Segmental Analysis
9.7.5 Africa
9.7.5.1 Africa Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.7.5.2 Market Size and Forecast
9.7.5.3 AfricaMarket Segmental Analysis
9.7.6 Brazil
9.7.6.1 Brazil Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.7.6.2 Market Size and Forecast
9.7.6.3 BrazilMarket Segmental Analysis
9.7.7 Rest of LAMEA
9.7.7.1 Rest of LAMEA Obesity Clinical Trials Market Revenue, 2022-2034 ($Billion)
9.7.7.2 Market Size and Forecast
9.7.7.3 Rest of LAMEAMarket Segmental Analysis
Chapter 10. Competitive Landscape
10.1 Competitor Strategic Analysis
10.1.1 Top Player Positioning/Market Share Analysis
10.1.2 Top Winning Strategies, By Company, 2022-2024
10.1.3 Competitive Analysis By Revenue, 2022-2024
10.2 Recent Developments by the Market Contributors (2024)
Chapter 11. Company Profiles
11.1 Novo Nordisk A/S
11.1.1 Company Snapshot
11.1.2 Company and Business Overview
11.1.3 Financial KPIs
11.1.4 Product/Service Portfolio
11.1.5 Strategic Growth
11.1.6 Global Footprints
11.1.7 Recent Development
11.1.8 SWOT Analysis
11.2 Eli Lilly and Company
11.3 Pfizer Inc.
11.4 Amgen Inc.
11.5 Altimmune, Inc.
11.6 Roche Holding AG
11.7 AstraZeneca plc
11.8 Boehringer Ingelheim International GmbH
11.9 Structure Therapeutics
11.10 Zealand Pharma A/S