The global robotics in cell therapy manufacturing market size reached USD 1.72 billion in 2025 and is expected to be worth around USD 6.91 billion by 2035, exhibiting a compound annual growth rate (CAGR) of 14.9% over the forecast period 2026 to 2035. Robotics adoption in cell therapy manufacturing is growing as manufacturers aim to scale complex, manual processes. Current methods are labor-intensive and rely on manual handling, increasing the risk of contamination, batch variability, and human error. The rising demand for advanced therapies, especially in oncology where cell-based treatments are expected to be vital, is creating a need for consistent, high-quality products at scale. Robotics and automated closed systems support this transition by enhancing standardization, sterility, and reproducibility, while also decreasing reliance on manual labor. Recent regulatory approvals and ongoing innovations further facilitate adoption. Between 2023 and mid-2024, about 10 cell and gene therapies received FDA approval, reflecting rapid pipeline growth and emphasising the necessity for scalable manufacturing solutions.
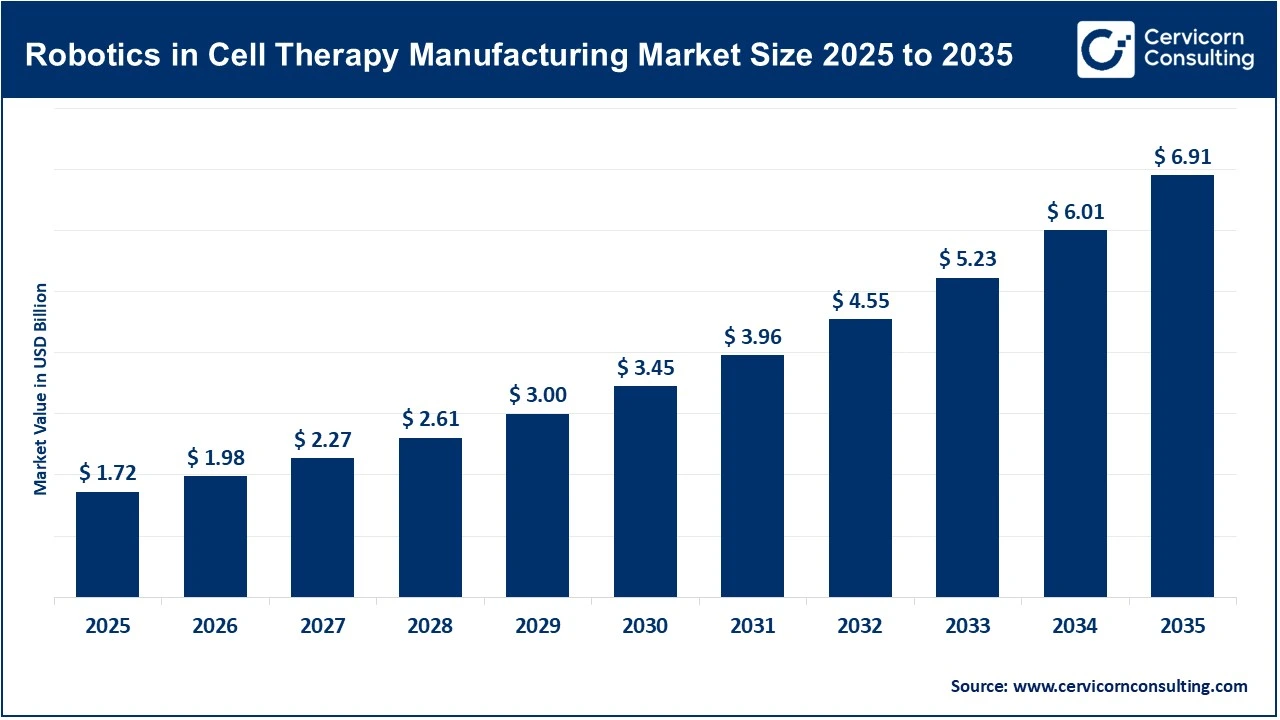
The integration of AI-driven robotics and advanced automation is emerging as a key factor in boosting efficiency and process control in cell therapy manufacturing. Robotic systems now automate entire workflows, from cell isolation to fill-finish, while AI enables predictive analytics, defect detection, and real-time decision-making. These advances have resulted in measurable improvements: biopharma organizations report up to 44% faster R&D outputs and around 30% cost savings, along with a significant reduction in manual errors. Industry investment in automated and closed systems is also rising, with the market expected to grow from approximately USD 1.72 billion in 2025 to over USD 6.91 billion by 2035. This trend indicates that robotics and automation are likely to become central to scaling and commercializing cell therapy manufacturing.
Shift Toward Fully Automated Closed-System Manufacturing Driving Market Growth
Adoption of fully automated closed-system manufacturing is increasing in the robotics market, driven by the need for improved sterility, efficiency, and scalability. Evidence indicates that closed systems reduce contamination and human error, resulting in higher product quality and consistency. By maintaining controlled environments, these systems help minimize contamination and support process uniformity, both of which are essential for regulatory compliance and patient safety. Automated closed platforms also support high-throughput production and have contributed to rapid industry growth, with advanced systems showing annual adoption rates above 20% as biopharma companies move toward robotic manufacturing. Recent collaborations between technology providers and biopharma firms illustrate how closed, automated platforms are being used to lower labor costs, increase throughput, and achieve contamination-free GMP production at commercial scale. These developments suggest that closed-system robotics is likely to play a central role in scaling cell therapy manufacturing worldwide.
Report Scope
| Area of Focus | Details |
| Market Size in 2026 | USD 1.98 Billion |
| Market Size in 2035 | USD 6.91 Billion |
| CAGR 2026 to 2035 | 14.90% |
| Dominant Region | North America |
| Fastest Growing Region | Asai-Pacific |
| Key Segments | Component, Cell Type, Manufacturing Process Stage, Robotics Type, End-User, Region |
| Key Companies | Multiply Labs, Cellular Origins, Cellares, Thermo Fisher Scientific, Cytiva, Charles River Laboratories, Lonza Group, Miltenyi Biotec, Fresenius Kabi, Astellas Pharma, Yaskawa Electric Corporation, Cellafa Biosciences |
1. Cellares Receives FDA Advanced Manufacturing Technology (AMT) Designation (2025)
In April 2025, the FDA granted Advanced Manufacturing Technology designation to Cellares’ Cell Shuttle robotic manufacturing platform. This designation allows for priority regulatory review and may shorten approval timelines for therapies produced with the system. Regulatory recognition of robotic manufacturing addresses a key barrier to adoption by reducing compliance risk. As a result, both emerging biotech firms and established pharmaceutical companies are likely to view robotic platforms as a more viable option for cell therapy production. The increased regulatory support suggests that end-to-end automated systems could see broader adoption, positioning robotics as an integral part of cell therapy manufacturing processes.
2. Cellares and Bristol Myers Squibb USD 380M Manufacturing Agreement (2024)
In 2024, Cellares entered into a USD 380 million global capacity reservation and supply agreement with Bristol Myers Squibb to manufacture CAR-T therapies using its robotic platform. This agreement reflects increasing industry interest in robotic manufacturing systems for advanced therapies. High-value contracts of this scale suggest that pharmaceutical companies are beginning to view robotics as a viable option for commercial production, rather than limiting adoption to pilot projects. The shift toward full-scale deployment is likely to support broader market expansion and provide a foundation for sustained revenue growth in robotic manufacturing platforms.
3. Multiply Labs Expands Collaboration with Thermo Fisher Scientific (2025)
In September 2025, Multiply Labs expanded its collaboration with Thermo Fisher Scientific to integrate robotic systems with automated cell processing technologies. This development reflects a broader shift toward ecosystem partnerships between robotics providers and bioprocessing companies, which is increasingly important for integrating automation into established manufacturing processes. The combination of robotics with GMP-compliant tools may help reduce implementation complexity and risk, supporting more efficient adoption. As a result, the industry is likely to see increased interest in modular robotic platforms, particularly as manufacturers look for scalable solutions that align with evolving automation standards.
4. Cellular Origins & Fresenius Kabi Robotic Platform Integration Milestone (2026)
In January 2026, Cellular Origins and Fresenius Kabi reached an initial integration milestone by connecting the Constellation robotic manufacturing platform with cell processing systems. This marks a practical step toward incorporating robotics into GMP manufacturing environments, which is likely to support the industrialization of cell and gene therapy production. Early results suggest the platform may reduce labor requirements by as much as 16 times, improve space utilization by over 30 times, and lower production costs by approximately 51%. These projected efficiencies are attracting investment and may encourage the industry to adopt larger-scale, robotics-enabled production models.
The robotics in cell therapy manufacturing market is segmented into component, cell type, manufacturing process stage, robotics type, end-user, and geography.
Hardware leads the market, as production relies on physical automation systems like robotic arms, bioreactors, and closed processing units. These systems are central to maintaining sterility and enabling scalable operations. Recent industry data indicates that nearly 59% of investments in advanced therapy manufacturing target commercial-scale automated infrastructure. This trend highlights the critical role of hardware in supporting efficient and reliable manufacturing processes.
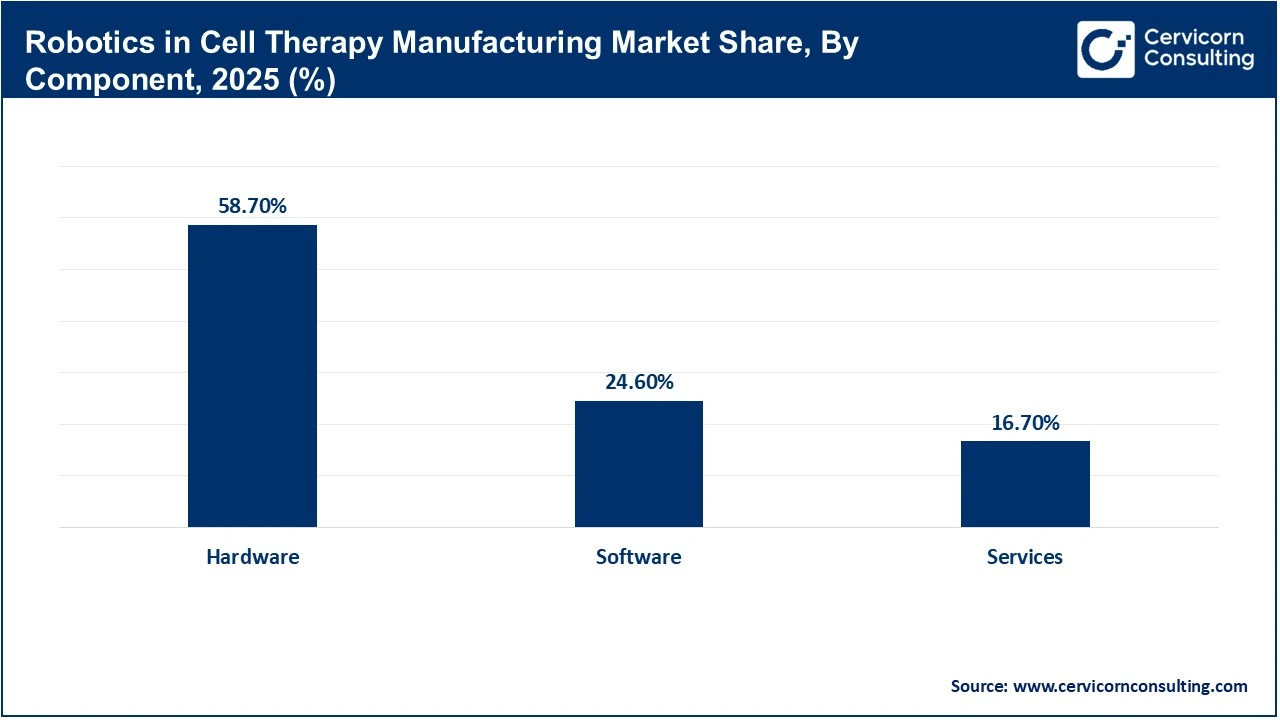
Software adoption in robotics is increasing as manufacturers integrate AI, digital twins, and manufacturing execution systems into their operations. This shift toward data-driven processes has led companies to implement software solutions for real-time monitoring, predictive analytics, and process optimization. The development of AI-enabled systems and advanced automation platforms is driving demand for software capable of managing complex workflows and supporting regulatory compliance. These trends suggest that software will play a central role in the evolving robotics ecosystem.
T-cell therapies continue to lead the market, primarily due to the commercial progress and regulatory approvals achieved by CAR-T therapies. The established manufacturing processes and broad clinical adoption of these therapies have positioned them as a significant component of current production pipelines. This leadership is supported by sustained investment and the development of supporting infrastructure. Recent data indicates that cell therapy manufacturing accounts for approximately 56% of advanced therapy production, with CAR-T and related modalities serving as the main drivers of this trend.
Robotics in Cell Therapy Manufacturing Market, By Cell Type, 2025 (%)
| Cell Type | Revenue Share, 2025 (%) |
| T-cells (CAR-T) | 52.30% |
| Stem Cells (MSC, iPSC) | 27.80% |
| Natural Killer (NK) Cells | 12.40% |
| Other Cell Types | 7.50% |
The stem cell segment is experiencing rapid growth, supported by expanding applications in regenerative medicine, neurology, and chronic disease treatment. The adoption of induced pluripotent stem cells (iPSCs) and the shift toward large-scale allogeneic manufacturing are increasing the need for automation and robotics. As stem cell therapies move toward high-volume, batch-based production, demand for robotic systems is expected to rise further. This trend suggests that stem cells will remain a key driver of automation adoption in the sector.
Downstream processing is currently the largest segment, encompassing cell expansion, harvesting, and purification. These are the most resource-intensive and critical steps in manufacturing, requiring high precision and strict contamination control. As a result, downstream operations are the main area for robotic automation. In automated workflows, cell separation alone accounts for over 32% of the market, underlining the significance of downstream processes in overall production.
Robotics in Cell Therapy Manufacturing Market, By Manufacturing Process Stage, 2025 (%)
| Manufacturing Process Stage | Revenue Share, 2025 (%) |
| Upstream Processing | 21.60% |
| Downstream Processing | 39.80% |
| Fill-Finish Operations | 14.70% |
| Quality Control (QC) | 23.90% |
Quality control is the fastest-growing segment, driven by stricter regulatory requirements and the need for real-time monitoring. As more therapies move into clinical and commercial production, manufacturers are adopting robotic quality control systems with AI-based analytics and sensors to maintain batch consistency and meet compliance standards. The shift toward automated and continuous monitoring is expected to further accelerate growth in this segment.
The market for fully automated closed systems is growing strongly, mainly because these systems enable end-to-end manufacturing with minimal human involvement. They help maintain sterility and reproducibility, which is critical for meeting GMP standards and reducing contamination risks. As a result, closed systems are becoming the preferred choice for commercial-scale production, with more facilities worldwide adopting this technology to support large-scale operations.
Robotics in Cell Therapy Manufacturing Market, By Robotics Type, 2025 (%)
| Robotics Type | Revenue Share, 2025 (%) |
| Industrial Robotics | 28.50% |
| Collaborative Robots (Cobots) | 16.20% |
| Fully Automated Closed Systems | 41.70% |
| AI-driven / Smart Robotics | 13.60% |
AI-driven robotics is emerging as the fastest-growing segment, mainly because it enables self-optimizing processes. By using machine learning for predictive maintenance, anomaly detection, and process optimization, these systems help improve efficiency and lower costs. As more companies integrate AI into their robotic platforms, manufacturing is shifting toward a more data-driven, autonomous approach, which is expected to accelerate adoption in the coming years.
Pharmaceutical and biotechnology companies currently hold the largest share, reflecting their role as primary developers and commercializers. Their significant investments in manufacturing infrastructure, combined with robust pipelines and access to funding, have positioned them at the forefront of the market. Regulatory approvals have further supported their leadership, suggesting that established industry players are likely to maintain a competitive advantage as the sector evolves.
Robotics in Cell Therapy Manufacturing Market, By End-User, 2025 (%)
| End-User | Revenue Share, 2025 (%) |
| Pharmaceutical Companies | 34.90% |
| Biotechnology Companies | 26.80% |
| Contract Manufacturing Organizations (CMOs) | 24.30% |
| Research Institutes | 8.10% |
| Academic Hospitals | 5.90% |
Contract manufacturing organizations represent the fastest-growing segment, reflecting a broader shift toward outsourcing among biotechnology firms. The appeal of CMOs lies in their ability to provide cost efficiency, regulatory expertise, and scalable infrastructure, which addresses the operational needs of companies seeking flexibility. Current estimates indicate that contract manufacturing accounts for approximately 65% of the market, and this share is expected to increase as demand for adaptable production solutions continues to rise. This trend may create new opportunities for specialized providers, while also introducing considerations around quality control and supply chain management.
The robotics in cell therapy manufacturing market is segmented by region into North America, Europe, Asia-Pacific, Latin America, and LAMEA. Here is a brief overview of each region:
The North America robotics in cell therapy manufacturing market size was valued at USD 0.66 billion in 2025 and is forecasted to hit around USD 2.67 billion by 2035.
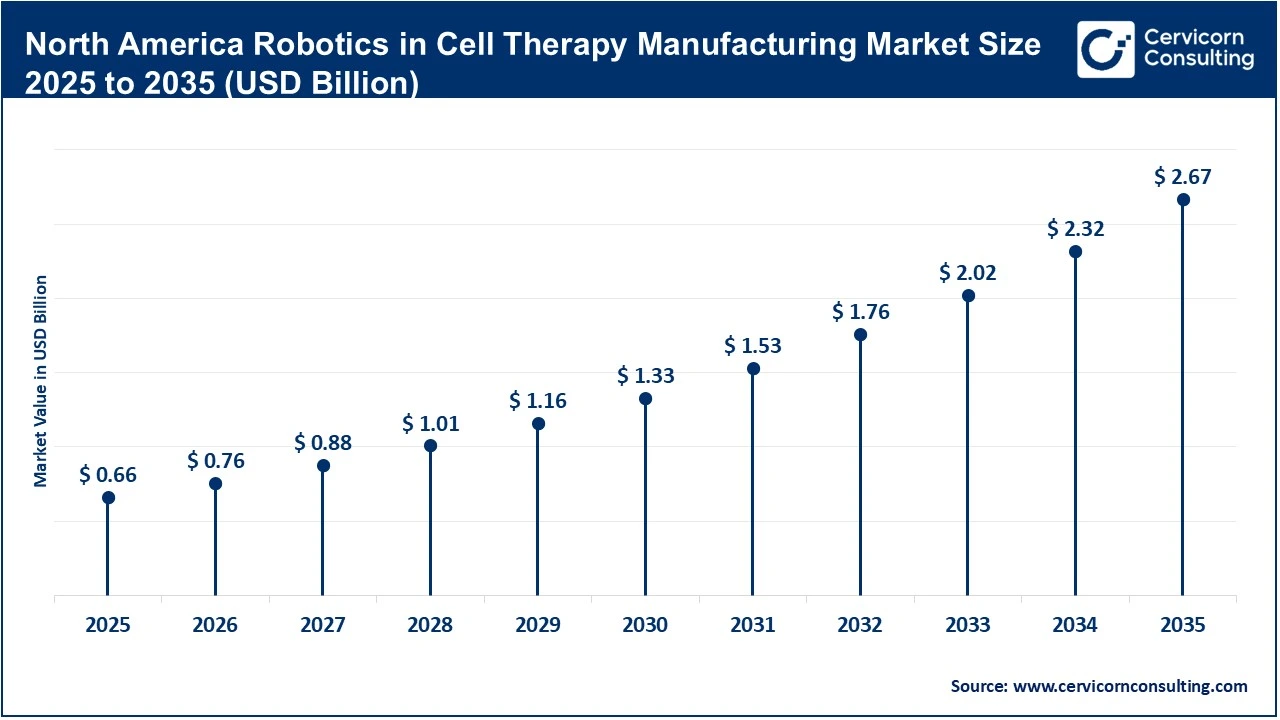
North America is the largest and most mature market, supported by strong biopharma innovation, regulatory support, and early adoption of robotics. The region benefits from high R&D spending and a well-established ecosystem of biotech firms, automation providers, and contract manufacturers. The presence of advanced manufacturing initiatives and regulatory encouragement for automated and closed systems is accelerating adoption. The region continues to lead in commercial-scale deployment of robotic platforms, making it the global benchmark for innovation.
United States: Leader in Commercialization and Advanced Manufacturing
Canada: Focus on Specialized Manufacturing and Research
The Europe robotics in cell therapy manufacturing market size was estimated at USD 0.47 billion in 2025 and is forecasted to grow around USD 1.89 billion by 2035. Europe is the second-largest market, characterized by strong regulatory oversight and collaborative research ecosystems. The region benefits from initiatives supporting advanced therapy medicinal products (ATMPs) and increasing investments in automation to meet strict quality standards. The adoption of robotics is growing as companies aim to improve process standardization and compliance. Additionally, public-private partnerships and funding programs are accelerating innovation in robotic manufacturing.
United Kingdom: Innovation in Automation and Advanced Therapies
Germany: Industrial Strength and Manufacturing Excellence
The Asia-Pacific robotics in cell therapy manufacturing market size reached USD 0.43 billion in 2025 and is expected to be worth around USD 1.71 billion by 2035. Asia-Pacific is the fastest-growing region, driven by rapid expansion of biopharma infrastructure and government-backed investments, accounting for approximately 25–30% of the global advanced therapy manufacturing ecosystem. Growth is fueled by rising demand for regenerative medicine and increased investments in automated production facilities. Countries across the region are expanding manufacturing capacity, with China and India experiencing double-digit annual growth (>15%) in biopharma manufacturing investment. The region’s cost advantage—offering 30–50% lower operational costs compared to Western markets—further accelerates the adoption of robotics and automation.
China: Government-Backed Expansion and Large-Scale Manufacturing
India: Cost Advantage and Emerging Biotech Ecosystem
Robotics in Cell Therapy Manufacturing Market Share, By Region, 2025 (%)
| Region | Revenue Share, 2025 (%) |
| North America | 38.60% |
| Europe | 27.40% |
| Asia-Pacific | 24.80% |
| LAMEA | 9.20% |
The LAMEA robotics in cell therapy manufacturing market was valued USD 0.16 billion in 2025 and is expected to be worth around USD 6.91 billion by 2035. Latin America holds a smaller share but is growing steadily due to improving healthcare infrastructure and rising adoption of advanced therapies. Countries like Brazil and Mexico are investing in biotechnology, with Brazil alone managing large-scale clinical research programs and expanding manufacturing capabilities. However, adoption of robotics remains limited due to cost and infrastructure constraints.
The Middle East & Africa region's growth is driven by government-led investments in healthcare and biotechnology. Countries such as the UAE and Saudi Arabia are investing heavily in biotech hubs and advanced manufacturing, with the UAE alone committing over USD 1–2 billion toward life sciences and innovation initiatives. These investments are gradually enabling the adoption of robotic manufacturing systems, although the region is still in an early development phase.
By Component
By Cell Type
By Manufacturing Process Stage
By Robotics Type
By End-User
By Geography