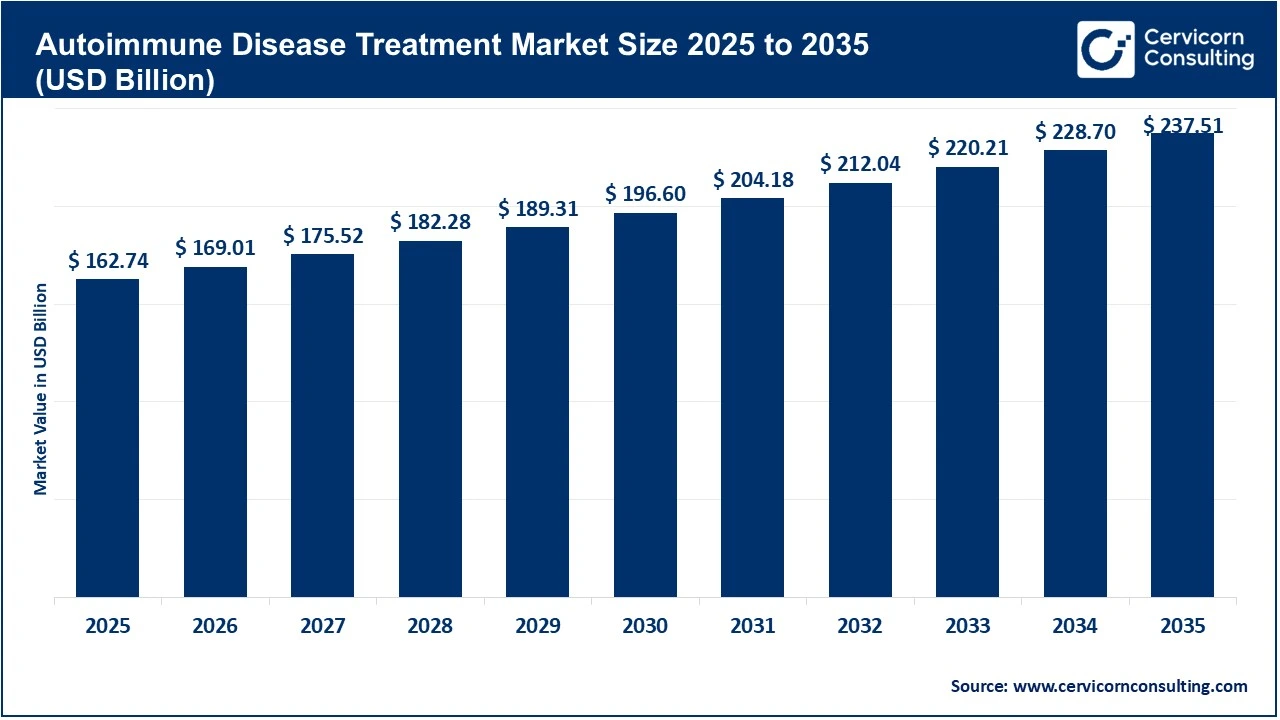
The global autoimmune disease treatment market size was valued at USD 162.74 billion in 2025 and is expected to be worth around USD 237.51 billion by 2035, exhibiting a compound annual growth rate (CAGR) of 3.9% over the forecast period from 2026 to 2035. The autoimmune disease treatment market is primarily driven by the rapidly increasing global disease burden and prevalence. Autoimmune disorders now affect around 1 in 10 people globally, with estimates indicating that 5–10% of populations in industrialised regions suffer from these conditions. More importantly, the epidemiological trend shows that global prevalence has nearly doubled between 1990 and 2021, while some studies indicate annual increases of about 12.5% in prevalence and 19.1% in incidence. This rising patient pool is fuelled by ageing populations, higher diagnosis rates, and environmental and lifestyle triggers such as processed diets, pollution, and changing immune exposures. Additionally, diseases like psoriasis alone affect over 125 million people globally, highlighting the scale of chronic demand for long-term therapies. As autoimmune conditions are typically lifelong and require continuous management, this expanding prevalence directly translates into sustained and recurring demand for therapeutics.
Another major growth driver is the shift towards advanced, targeted therapies and increasing R&D investments. The market is benefiting from the rapid adoption of biologics, immunomodulators, and precision therapies that offer higher efficacy than traditional treatments. Pharmaceutical innovation is accelerating, with growing investment in novel mechanisms such as cytokine inhibitors and next-generation immunotherapies, alongside emerging approaches, such as antigen-specific treatments, that aim for more precise immune modulation. At the same time, improved diagnosis and awareness are expanding the treated population millions of patients remain underdiagnosed, and earlier detection is increasing the rate of therapy initiation. Demographic factors also play a role: autoimmune diseases disproportionately affect women (up to 63% of cases) and are rising with age, particularly after 50, further expanding the addressable patient base. Together, these factors, rising prevalence, chronic treatment needs, therapeutic innovation, and improved diagnosis, are creating strong, sustained growth momentum in the autoimmune disease treatment market.
Rising Disease Burden and Advanced Targeted Therapies Driving Market Growth
The autoimmune disease treatment market is being strongly driven by the rising prevalence of autoimmune conditions combined with a shift toward more effective, targeted therapies. Globally, autoimmune diseases affect an estimated 5–10% of the population, and the number of diagnosed cases continues to increase due to better screening, urban lifestyles, environmental triggers, and aging demographics. Since most autoimmune disorders, such as rheumatoid arthritis, psoriasis, and multiple sclerosis, are chronic and require lifelong management, each new diagnosis adds long-term demand for therapies rather than one-time treatment. At the same time, earlier diagnosis is expanding the treated population, while higher disease awareness is pushing more patients into formal healthcare systems. This growing patient pool, combined with the need for continuous medication, creates a compounding demand effect, which is further amplified by the transition from traditional drugs to high-value advanced therapies that improve outcomes but also increase treatment adoption rates.
Report Scope
| Area of Focus | Details |
| Market Size in 2026 | USD 169.01 Billion |
| Market Size in 2035 | USD 237.51 Billion |
| CAGR 2026 to 2035 | 3.90% |
| Dominant Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Key Segments | Drug Class, Mechanism of Action, Disease Indication, Route of Administration, End User |
| Key Companies | AbbVie Inc., Amgen Inc., Johnson & Johnson, F. Hoffmann-La Roche AG, Pfizer Inc., AstraZeneca plc, Bristol-Myers Squibb Company, Eli Lilly and Company, Novartis AG, Sanofi S.A., GlaxoSmithKline plc, UCB S.A. |
1. Record Growth in Biosimilar Approvals (FDA – 2024–2025)
The U.S. Food and Drug Administration approved a record 18 biosimilars in 2024, the highest ever, significantly expanding access to autoimmune treatments such as monoclonal antibodies.
This milestone is driving the market by reducing treatment costs and increasing accessibility, especially for chronic conditions like rheumatoid arthritis and inflammatory bowel disease that rely on expensive biologics. As biosimilars enter the market, they intensify competition, improve affordability, and expand patient coverage in both developed and emerging regions, thereby increasing overall treatment penetration and volume growth.
2. Expansion of Biologic Indications (Dupilumab Approval – 2025)
Dupilumab received new approvals in 2025 for additional autoimmune-related conditions, including chronic spontaneous urticaria and bullous pemphigoid.
This milestone demonstrates how existing biologics are being repurposed across multiple indications, allowing pharmaceutical companies to maximize the value of a single drug across several diseases. This drives the market by expanding the treatable patient population without requiring entirely new drug development, accelerating revenue growth and reinforcing the dominance of biologics in autoimmune therapy.
3. Pipeline Breakthroughs and First-in-Class Treatments (Johnson & Johnson – 2026)
Johnson & Johnson is seeking FDA approval for nipocalimab as the first-ever treatment for warm autoimmune hemolytic anemia, a rare but serious disease with no approved therapies.
This milestone is significant because it addresses high unmet medical needs in rare autoimmune diseases, opening entirely new market segments. First-in-class therapies command premium pricing and create new standards of care, thereby expanding the total addressable market and driving innovation-led growth within the industry.
4. Emergence of Cell & Gene Therapies (CAR-T for Autoimmune Diseases – 2026)
Regulatory progress, including FDA-cleared trials for CAR-T therapies, highlights their potential to “reset” the immune system and potentially cure autoimmune diseases rather than just manage them.
This milestone is transformative as it shifts the market from chronic symptom management to potential one-time curative treatments, which can dramatically improve patient outcomes and reduce long-term dependency on drugs. At the same time, it attracts substantial investment in advanced therapeutics, accelerating R&D and reshaping the competitive landscape toward high-value, next-generation therapies.
The autoimmune disease treatment market is segmented by region into North America, Europe, Asia-Pacific, Latin America, and LAMEA. Here is a brief overview of each region:
The North America autoimmune disease treatment market size was valued at USD 162.74 billion in 2025 and is expected to be worth around USD 237.51 billion by 2035.
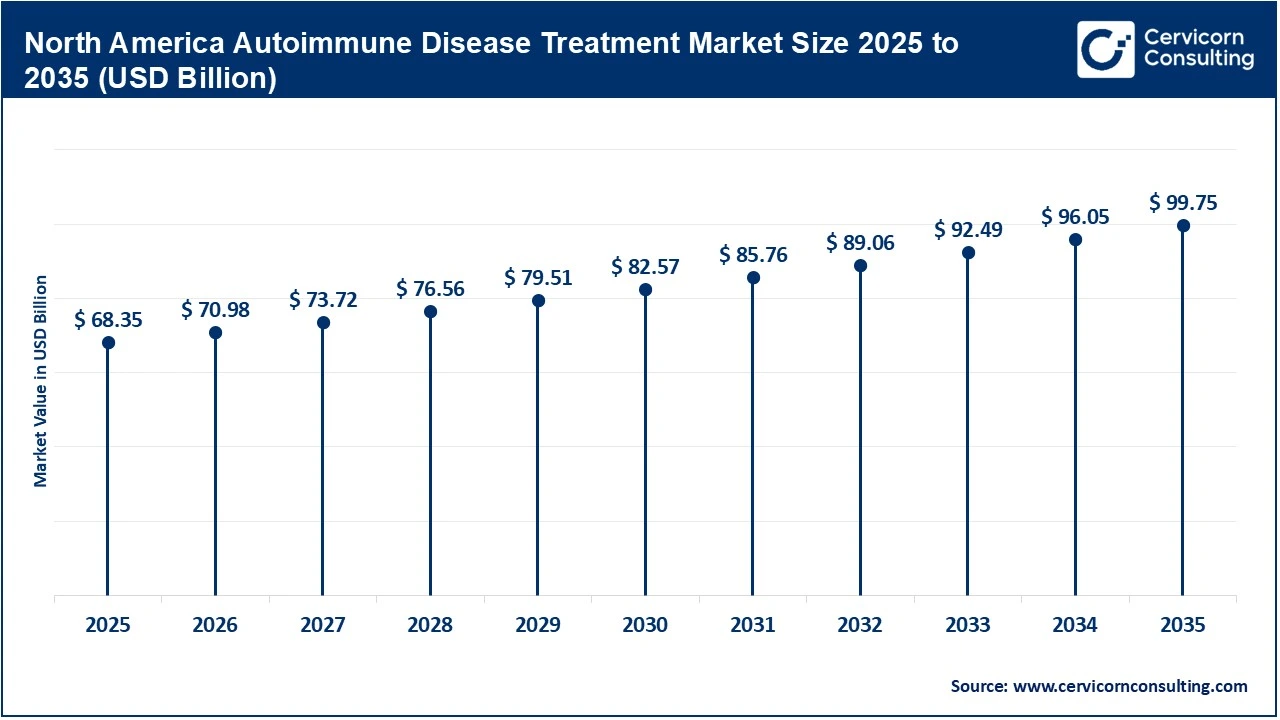
The North America market is highly developed, supported by advanced healthcare systems, strong reimbursement frameworks, and early adoption of innovative therapies. The region leads in biologics and targeted immunotherapies due to high healthcare spending and the strong presence of major pharmaceutical companies. Additionally, widespread awareness, early diagnosis, and access to specialty care significantly increase treatment rates. Continuous R&D investments, favorable regulatory policies, and rapid uptake of advanced therapies further strengthen market growth, making North America the most mature and innovation-driven region globally.
United States: High biologics adoption, strong R&D ecosystem, and favorable reimbursement policies are significantly driving market growth across the country.
Canada: Strong public healthcare system, increasing biosimilar adoption, and supportive government initiatives are steadily expanding market access.
The Asia-Pacific autoimmune disease treatment market size was valued at USD 162.74 billion in 2025 and is expected to be worth around USD 237.51 billion by 2035. The Asia-Pacific market is experiencing rapid growth, driven by a large, underdiagnosed patient population, improving healthcare infrastructure, and rising disease awareness. Increasing urbanization, environmental factors, and lifestyle changes are contributing to higher incidence of autoimmune conditions. Governments are expanding healthcare coverage and investing in medical infrastructure, while pharmaceutical companies are strengthening their regional presence. The growing availability of biosimilars and cost-effective therapies is significantly improving access, positioning Asia-Pacific as the fastest-growing market globally.
China: Expanding healthcare access, strong domestic biologics manufacturing capabilities, and increasing government support for innovative therapies are significantly driving market growth.
India: Growing awareness, improving healthcare infrastructure, and strong adoption of cost-effective therapies are accelerating market expansion.
The Europe autoimmune disease treatment market size was valued at USD 162.74 billion in 2025 and is expected to be worth around USD 237.51 billion by 2035. The Europe market is well established, driven by universal healthcare coverage, structured treatment guidelines, and strong regulatory frameworks. Governments emphasize cost optimization, which has significantly accelerated biosimilar adoption and improved patient access to advanced therapies. The region benefits from high disease awareness, strong pharmaceutical manufacturing capabilities, and consistent reimbursement support. Additionally, ongoing investments in research and cross-border healthcare collaboration are enhancing innovation and access, making Europe a stable and cost-efficient market for autoimmune disease treatments.
Germany: Strong pharmaceutical industry presence, high biologics usage, and advanced healthcare infrastructure are driving sustained market growth.
United Kingdom: Government-led healthcare system, strong biosimilar uptake, and early diagnosis initiatives are supporting market expansion.
Autoimmune Disease Treatment Market Share, By Region, 2025 (%)
| Region | Revenue Share, 2025 (%) |
| North America | 42%42% |
| Europe | 28% |
| Asia-Pacific | 22% |
| LAMEA | 8% |
The LAMEA autoimmune disease treatment market size was valued at USD 162.74 billion in 2025 and is expected to be worth around USD 237.51 billion by 2035. The Latin America market is developing steadily, driven by improving healthcare infrastructure, rising disease awareness, and gradual adoption of advanced therapies. While high treatment costs limit widespread use of biologics, increasing availability of biosimilars is helping bridge the affordability gap. Governments are investing in healthcare reforms and expanding insurance coverage, which is improving access to diagnosis and treatment.
The Middle East & Africa autoimmune disease treatment market is in a developing stage, supported by increasing healthcare investments, improving infrastructure, and rising awareness of autoimmune conditions. While affordability and access challenges persist, government initiatives and private-sector involvement are gradually improving treatment availability. The region is expected to witness steady growth as healthcare systems continue to evolve and expand.
Brazil: Expanding public healthcare coverage, increasing biosimilar adoption, and rising disease awareness are driving market growth.
Saudi Arabia: Government-led healthcare transformation programs, increasing investments, and growing adoption of advanced therapies are driving market growth.
The autoimmune disease treatment market is segmented into drug class, mechanism of action, disease indication, route of administration, end-user, and geography.
Biologics dominate the autoimmune disease treatment market due to their superior efficacy in targeting specific immune pathways such as TNF and interleukins. These therapies are widely prescribed for chronic conditions like rheumatoid arthritis, psoriasis, and inflammatory bowel disease, where conventional drugs often fail to deliver sustained results. Their ability to provide long-term disease control and remission has led to strong physician preference. Additionally, continuous innovation, brand loyalty, and extensive clinical validation have reinforced their leadership, making biologics the largest revenue-generating segment globally.
Autoimmune Disease Treatment Market Share, By Drug Class, 2025 (%)
| Drug Class | Revenue Share, 2025 (%) |
| Biologics | 55% |
| Biosimilars | 10% |
| Small Molecule Drugs | 12% |
| Immunosuppressants | 8% |
| Corticosteroids | 6% |
| Nonsteroidal Anti-inflammatory Drugs (NSAIDs) | 5% |
| Others | 4% |
Biosimilars are emerging as the fastest-growing segment, driven by their cost-effectiveness and increasing regulatory approvals worldwide. As patents for major biologics expire, biosimilars provide affordable alternatives with comparable safety and efficacy, making advanced treatments accessible to a broader patient population. This is particularly significant in emerging markets where high biologic costs limit adoption. Government support for cost containment and healthcare expansion is further accelerating uptake. As competition intensifies, biosimilars are expected to significantly increase treatment penetration and reshape pricing dynamics in the market.
TNF inhibitors dominate the mechanism of action segment due to their long-standing clinical success and widespread use in treating multiple autoimmune diseases. These drugs were among the first targeted biologics and remain the standard of care for conditions like rheumatoid arthritis, psoriasis, and Crohn’s disease. Their proven efficacy, extensive safety data, and strong physician familiarity have sustained their leading position. Despite increasing competition from newer therapies, TNF inhibitors continue to generate substantial demand due to their broad applicability and well-established treatment protocols.
Autoimmune Disease Treatment Market, By Mechanism of Action, 2025 (%)
| Mechanism of Action | Revenue Share, 2025 (%) |
| TNF Inhibitors | 35% |
| Interleukin Inhibitors (IL-6, IL-17, IL-23) | 22% |
| Janus Kinase (JAK) Inhibitors | 12% |
| B-cell Inhibitors | 10% |
| T-cell Inhibitors | 8% |
| Phosphodiesterase-4 (PDE4) Inhibitors | 6% |
| Others | 7% |
JAK inhibitors are the fastest-growing segment owing to their oral administration, rapid onset of action, and effectiveness in patients who do not respond to traditional biologics. These targeted small-molecule therapies offer convenience and flexibility, improving patient adherence compared to injectable treatments. Increasing approvals and expanding indications across multiple autoimmune diseases are further boosting their adoption. Additionally, ongoing research and development are enhancing their safety profiles, positioning JAK inhibitors as a strong alternative to biologics and driving rapid growth within this segment.
Rheumatoid arthritis dominates the disease indication segment due to its high global prevalence and chronic nature requiring long-term treatment. It accounts for a significant proportion of autoimmune therapy demand, supported by strong clinical awareness and established treatment guidelines. The availability of multiple therapeutic options, including biologics and targeted therapies, further drives market share. Additionally, early diagnosis initiatives and increasing healthcare access have expanded the treated population, reinforcing rheumatoid arthritis as the leading contributor to overall market revenue.
Autoimmune Disease Treatment Market, By Disease Indication, 2025 (%)
| Disease Indication | Revenue Share, 2025 (%) |
| Rheumatoid Arthritis | 30% |
| Psoriasis | 18% |
| Multiple Sclerosis | 15% |
| Inflammatory Bowel Disease (IBD) | 14% |
| Systemic Lupus Erythematosus (SLE) | 8% |
| Type 1 Diabetes | 6% |
| Ankylosing Spondylitis | 5% |
| Others | 4% |
Inflammatory bowel disease is the fastest-growing segment due to rising incidence rates, particularly in urbanized and developing regions. Lifestyle changes, dietary factors, and environmental influences are contributing to increased diagnosis of Crohn’s disease and ulcerative colitis. Advances in biologics and targeted therapies have significantly improved treatment outcomes, encouraging higher adoption rates. Moreover, growing awareness and improved diagnostic capabilities are enabling earlier intervention, which is expanding the patient pool and accelerating growth within the IBD segment.
Injectable therapies dominate the route of administration segment as most biologics, which are the leading treatment class, are delivered via subcutaneous or intravenous routes. These methods ensure high bioavailability and precise targeting of immune pathways, making them highly effective for severe autoimmune conditions. Hospitals and specialty clinics are well-equipped to administer these therapies, further supporting their widespread use. Despite the inconvenience compared to oral drugs, their superior clinical outcomes and established treatment protocols maintain their dominance in the market.
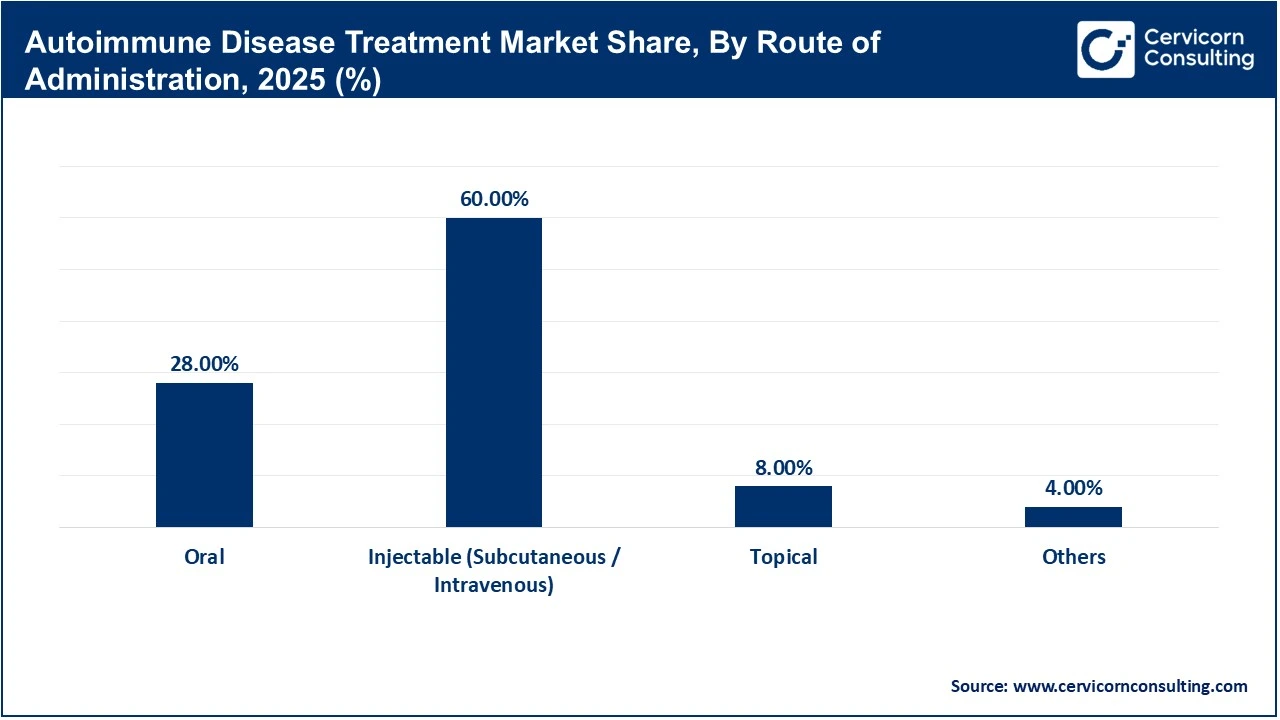
Oral therapies are the fastest-growing segment due to their convenience, ease of administration, and improved patient compliance. Drugs such as JAK inhibitors have demonstrated strong efficacy while eliminating the need for injections, making them highly attractive to patients and physicians. Increasing preference for self-administered treatments, especially in homecare settings, is further driving growth. Additionally, ongoing innovation in oral small-molecule drugs is expanding treatment options, positioning this segment for rapid adoption across multiple autoimmune conditions.
Hospitals dominate the end-user segment due to their central role in diagnosing and managing complex autoimmune diseases. They provide access to advanced therapies, including biologics and infusion-based treatments, along with specialized medical expertise. Severe and newly diagnosed cases are typically managed in hospital settings, ensuring consistent patient inflow. Additionally, strong infrastructure, reimbursement support, and multidisciplinary care approaches contribute to their leading position in the market.
Autoimmune Disease Treatment Market, By End User, 2025 (%)
| End User | Revenue Share, 2025 (%) |
| Hospitals | 45% |
| Specialty Clinics | 30% |
| Homecare Settings | 20% |
| Research Institutes | 5% |
Homecare settings are the fastest-growing segment as healthcare systems increasingly shift toward patient-centric and cost-effective care models. The availability of self-administered therapies, such as subcutaneous biologics and oral drugs, enables patients to manage chronic conditions at home. This reduces hospital visits and overall treatment costs while improving convenience and quality of life. Growing digital health support and remote monitoring technologies are further facilitating this transition, driving rapid growth in the homecare segment.
By Drug Class
By Mechanism of Action
By Disease Indication
By Route of Administration
By End User
By Geography